Zelatriazin
Appearance
 | |
| Clinical data | |
|---|---|
| Other names | NBI-1065846; TAK-041 |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| Chemical and physical data | |
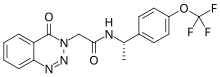
| Formula | C18H15F3N4O3 |
| Molar mass | 392.338 g·mol−1 |
Zelatriazin (NBI-1065846 or TAK-041) is a small-molecule agonist of GPR139. It was developed for schizophrenia and anhedonia in depression but trials were unsuccessful and its development was discontinued in 2023.[1][2][3][4][5][6][7]
References
[edit]- ^ Kamel, Amin; Bowlin, Steve; Hosea, Natalie; Arkilo, Dimitrios; Laurenza, Antonio (February 2021). "In Vitro Metabolism of Slowly Cleared G Protein–Coupled Receptor 139 Agonist TAK-041 Using Rat, Dog, Monkey, and Human Hepatocyte Models (HepatoPac): Correlation with In Vivo Metabolism". Drug Metabolism and Disposition. 49 (2): 121–132. doi:10.1124/dmd.120.000246. PMID 33273044. S2CID 227282766.
- ^ Schiffer, Hans; Atienza, Josephine; Reichard, Holly; Mulligan, Victoria; Cilia, Jackie; Monenschein, Holger; Collia, Deanna; Ray, Jim; Kilpatrick, Gavin; Brice, Nicola; Carlton, Mark; Hitchcock, Steve; Corbett, Ged; Hodgson, Robert (18 May 2020). "S180. The Selective Gpr139 Agonist Tak-041 Reverses Anhedonia and Social Interaction Deficits in Rodent Models Related to Negative Symptoms in Schizophrenia". Schizophrenia Bulletin. 46 (Supplement_1): S106 – S107. doi:10.1093/schbul/sbaa031.246. PMC 7234360.
- ^ Yin, Wei; Han, David; Khudyakov, Polyna; Behrje, Rhett; Posener, Joel; Laurenza, Antonio; Arkilo, Dimitrios (August 2022). "A phase 1 study to evaluate the safety, tolerability and pharmacokinetics of TAK-041 in healthy participants and patients with stable schizophrenia". British Journal of Clinical Pharmacology. 88 (8): 3872–3882. doi:10.1111/bcp.15305. PMC 9544063. PMID 35277995. S2CID 247407736.
- ^ Rabiner, Eugenii A.; Uz, Tolga; Mansur, Ayla; Brown, Terry; Chen, Grace; Wu, Jingtao; Atienza, Joy; Schwarz, Adam J.; Yin, Wei; Lewis, Yvonne; Searle, Graham E.; Dennison, Jeremy M. T. J.; Passchier, Jan; Gunn, Roger N.; Tauscher, Johannes (June 2022). "Endogenous dopamine release in the human brain as a pharmacodynamic biomarker: evaluation of the new GPR139 agonist TAK-041 with [11C]PHNO PET". Neuropsychopharmacology. 47 (7): 1405–1412. doi:10.1038/s41386-021-01204-1. PMC 9117280. PMID 34675381.
- ^ Reichard, Holly A.; Schiffer, Hans H.; Monenschein, Holger; Atienza, Josephine M.; Corbett, Gerard; Skaggs, Alton W.; Collia, Deanna R.; Ray, William J.; Serrats, Jordi; Bliesath, Joshua; Kaushal, Nidhi; Lam, Betty P.; Amador-Arjona, Alejandro; Rahbaek, Lisa; McConn, Donavon J.; Mulligan, Victoria J.; Brice, Nicola; Gaskin, Philip L. R.; Cilia, Jackie; Hitchcock, Stephen (12 August 2021). "Discovery of TAK-041: a Potent and Selective GPR139 Agonist Explored for the Treatment of Negative Symptoms Associated with Schizophrenia". Journal of Medicinal Chemistry. 64 (15): 11527–11542. doi:10.1021/acs.jmedchem.1c00820. PMID 34260228. S2CID 235908256.
- ^ Münster, Alexandra; Sommer, Susanne; Kúkeľová, Diana; Sigrist, Hannes; Koros, Eliza; Deiana, Serena; Klinder, Klaus; Baader-Pagler, Tamara; Mayer-Wrangowski, Svenja; Ferger, Boris; Bretschneider, Tom; Pryce, Christopher R.; Hauber, Wolfgang; von Heimendahl, Moritz (August 2022). "Effects of GPR139 agonism on effort expenditure for food reward in rodent models: Evidence for pro-motivational actions". Neuropharmacology. 213: 109078. doi:10.1016/j.neuropharm.2022.109078. PMID 35561791. S2CID 248574904.
- ^ Taylor, Nick Paul (10 November 2023). "Neurocrine hit with one-two punch as Takeda and Xenon pacts deliver midphase flops". Fierce Biotech. Retrieved 4 December 2023.
