User:Tobias1984/Infobox drug test
| This page in a nutshell: This is just a copy of a Wikipedia entry to experiment with the infobox layout |
| Clinical data | |
|---|---|
| License data | |
| ../licence/sandbox | |
| License data | |
Warfarin (also known by the brand names Coumadin, Jantoven, Marevan, Uniwarfin) is an anticoagulant normally used in the prevention of thrombosis and thromboembolism, the formation of blood clots in the blood vessels and their migration elsewhere in the body, respectively. Warfarin is commonly but incorrectly referred to as a blood thinner. It was initially introduced in 1948 as a pesticide against rats and mice, and is still used for this purpose, although more potent poisons such as brodifacoum have since been developed. In the early 1950s, warfarin was found to be effective and relatively safe for preventing thrombosis and thromboembolism in many disorders. It was approved for use as a medication in 1954, and has remained popular ever since; warfarin is the most widely prescribed oral anticoagulant drug in North America.[1]
Despite its effectiveness, treatment with warfarin has several shortcomings. Many commonly used medications interact with warfarin, as do some foods (particularly leaf vegetable foods or "greens," since these typically contain large amounts of vitamin K1) and its activity has to be monitored by blood testing for the international normalized ratio (INR) to ensure an adequate yet safe dose is taken.[2] A high INR predisposes to a high risk of bleeding, while an INR below the therapeutic target indicates the dose of warfarin is insufficient to protect against thromboembolic events.
Warfarin and related 4-hydroxycoumarin-containing molecules decrease blood coagulation by inhibiting vitamin K epoxide reductase, an enzyme that recycles oxidized vitamin K1 to its reduced form after it has participated in the carboxylation of several blood coagulation proteins, mainly prothrombin and factor VII. Despite being labeled a vitamin K antagonist,[2] warfarin does not antagonize the action of vitamin K1, but rather antagonizes vitamin K1 recycling, depleting active vitamin K1.[3] Thus, the pharmacologic action may always be reversed by fresh vitamin K1. When administered, these drugs do not anticoagulate blood immediately. Instead, onset of their effect requires about a day before remaining active clotting factors have had time to naturally disappear in metabolism, and the duration of action of a single dose of warfarin is 2 to 5 days. Reversal of warfarin's effect when it is discontinued or vitamin K1 is administered, requires a similar time.
Warfarin is a synthetic derivative of dicoumarol, a 4-hydroxycoumarin-derived mycotoxin anticoagulant originally discovered in spoiled sweet clover-based animal feeds. Dicoumarol, in turn, is derived from coumarin, a sweet-smelling but coagulation-inactive chemical found naturally in "sweet" clover (to which it gives its odor and name), tonka beans (also known as "cumaru" from which coumarin's name derives), and many other plants. The name 'warfarin' stems from its discovery at the University of Wisconsin, incorporating the acronym for the organization that funded the key research, "WARF" for the Wisconsin Alumni Research Foundation and the ending "-arin", indicating its link with coumarin.
Medical uses
[edit]Warfarin is used to decrease the tendency for thrombosis or as secondary prophylaxis (prevention of further episodes) in those individuals who have already formed a blood clot (thrombus). Warfarin treatment can help prevent formation of future blood clots and help reduce the risk of embolism (migration of a thrombus to a spot where it blocks blood supply to a vital organ).[4]
Warfarin is best suited for anticoagulation (clot formation inhibition) in areas of slowly running blood (such as in veins and the pooled blood behind artificial and natural valves) and in blood pooled in dysfunctional cardiac atria. Thus, common clinical indications for warfarin use are atrial fibrillation, the presence of artificial heart valves, deep venous thrombosis, and pulmonary embolism (where the embolized clots first form in veins). Warfarin is also used in antiphospholipid syndrome. It has been used occasionally after heart attacks (myocardial infarctions), but is far less effective at preventing new thromboses in coronary arteries. Prevention of clotting in arteries is usually undertaken with antiplatelet drugs, which act by a different mechanism from warfarin (which normally has no effect on platelet function).[5]
Alternative anticoagulants
[edit]In some countries, other coumarins are used instead of warfarin, such as acenocoumarol and phenprocoumon. These have a shorter (acenocoumarol) or longer (phenprocoumon) half-life, and are not completely interchangeable with warfarin. Several types of anticoagulant drugs offering the efficacy of warfarin without a need for monitoring, such as dabigatran and rivaroxaban, have been approved in a number of countries for classical warfarin uses like the more common types of atrial fibrillation, and others in the same drug classes are under development.[6]
Dosing
[edit]Dosing of warfarin is complicated because it is known to interact with many commonly used medications and certain foods.[1] These interactions may enhance or reduce warfarin's anticoagulation effect. To optimize the therapeutic effect without risking dangerous side effects such as bleeding, close monitoring of the degree of anticoagulation is required by a blood test measuring an INR. During the initial stage of treatment, INR is checked daily; intervals between tests can be lengthened if the patient manages stable therapeutic INR levels on an unchanged warfarin dose.[5] Newer point-of-care testing is available and has increased the ease of INR testing in the outpatient setting. Instead of a blood draw, the point of care test involves a simple finger prick.[7]
When initiating warfarin therapy ("warfarinization"), the doctor will decide how strong the anticoagulant therapy needs to be. The target INR level varies from case to case depending on the clinical indicators, but tends to be 2–3 in most conditions. In particular, target INR may be 2.5–3.5 (or even 3.0–4.5) in patients with one or more mechanical heart valves.[8]
In addition, for the first three days of "warfarinization", the levels of protein C and protein S (anticoagulation factors) drop faster than procoagulation proteins such as factor II, VII, IX, and X. Therefore, bridging anticoagulant therapies (usually heparin) are often used to reverse this temporary hypercoagulable state.
Maintenance dose
[edit]
Recommendations by many national bodies, including the American College of Chest Physicians,[10] have been distilled to help manage dose adjustments.[11]
The maintenance dose of warfarin can fluctuate significantly depending on the amount of vitamin K1 in the diet. Keeping vitamin K1 intake at a stable level can prevent these fluctuations. Leafy green vegetables tend to contain higher amounts of vitamin K1. Green parts of members of the family Apiaceae, such as parsley, cilantro, and dill, are extremely rich sources of vitamin K; cruciferous vegetables such as cabbage and broccoli, as well as the darker varieties of lettuces and other leafy greens, are also relatively high in vitamin K1. Green vegetables such a peas and green beans do not have such high amounts of vitamin K1 as leafy greens. Certain vegetable oils have high amounts of vitamin K1. Foods low in vitamin K1 include roots, bulbs, tubers, and most fruits and fruit juices. Cereals, grains and other milled products are also low in vitamin K1.[12]
Self-testing
[edit]Patients are making increasing use of self-testing and home monitoring of oral anticoagulation. International guidelines on home testing were published in 2005.[13] The guidelines stated: "The consensus agrees that patient self-testing and patient self-management are effective methods of monitoring oral anticoagulation therapy, providing outcomes at least as good as, and possibly better than, those achieved with an anticoagulation clinic. All patients must be appropriately selected and trained. Currently available self-testing/self-management devices give INR results that are comparable with those obtained in laboratory testing."[13] A 2006 systematic review and meta-analysis of 14 randomized trials showed home testing led to a reduced incidence of complications (thrombosis and major bleeding) and improved the time in the therapeutic range.[14]
Contraindications
[edit]Warfarin is contraindicated in pregnancy, as it passes through the placental barrier and may cause bleeding in the foetus; warfarin use during pregnancy is commonly associated with spontaneous abortion, stillbirth, neonatal death, and preterm birth.[15] Coumarins (such as warfarin) are also teratogens, that is, they cause birth defects; the incidence of birth defects in infants exposed to warfarin in utero appears to be around 5%, although higher figures (up to 30%) have been reported in some studies.[16] Depending on when exposure occurs during pregnancy, two distinct combinations of congenital abnormalities can arise.[15]
First trimester of pregnancy
[edit]Usually, warfarin is avoided in the first trimester, and a low molecular weight heparin such as enoxaparin is substituted. With heparin, risk of maternal haemorrhage and other complications are still increased, but heparins do not cross the placental barrier, so do not cause birth defects.[16] Various solutions exist for the time around delivery.
When warfarin (or another 4-hydroxycoumarin derivative) is given during the first trimester—particularly between the sixth and ninth weeks of pregnancy—a constellation of birth defects known variously as fetal warfarin syndrome (FWS), warfarin embryopathy, or coumarin embryopathy can occur. FWS is characterized mainly by skeletal abnormalities, which include nasal hypoplasia, a depressed or narrowed nasal bridge, scoliosis, and calcifications in the vertebral column, femur, and heel bone, which show a peculiar stippled appearance on X-rays. Limb abnormalities, such asbrachydactyly (unusually short fingers and toes) or underdeveloped extremities, can also occur.[15][16] Common nonskeletal features of FWS include low birth weight and developmental disabilities.[15][16]
Second trimester and later
[edit]Warfarin administration in the second and third trimesters is much less commonly associated with birth defects, and when they do occur, are considerably different from fetal warfarin syndrome. The most common congenital abnormalities associated with warfarin use in late pregnancy are central nervous system disorders, including spasticity and seizures, and eye defects.[15][16] Because of such later pregnancy birth defects, anticoagulation with warfarin poses a problem in pregnant women requiring warfarin for vital indications, such as stroke prevention in those with artificial heart valves.
According to the American College of Chest Physicians (ACCP), warfarin may be used in lactating women who wish to breast-feed their infants.[17] Available data does not suggest that warfarin crosses into the breast milk. Similarly, INR levels should be checked to avoid adverse effects.[17]
Adverse effects
[edit]Hemorrhage
[edit]The only common side effect of warfarin is haemorrhage (bleeding). The risk of severe bleeding is small but definite (a median annual rate of 1 to 3% has been reported[10]) and any benefit needs to outweigh this risk when warfarin is considered as a therapeutic measure. All types of bleeding occur more commonly, but the most catastrophic ones are those involving the brain (intracerebral hemorrhage/hemorrhagic stroke) and the spinal cord.[10] Risk of bleeding is increased if the INR is out of range (due to accidental or deliberate overdose or due to interactions).[18]
A number of risk scores exists to predict bleeding in people using warfarin and similar anticoagulants. A commonly used score (HAS-BLED) includes known predictors of warfarin-related bleeding: uncontrolled high blood pressure (H), abnormal kidney function (A), previous stroke (S), known previous bleeding condition (B), previous labile INR when on anticoagulation (L), elderly as defined by age over 65 (E), and drugs associated with bleeding (e.g. aspirin) or alcohol misuse (D). While their use is recommended in clinical practice guidelines,[19] they are only moderately effective in predicting bleeding risk and don't perform well in predicting hemorrhagic stroke.[20] Bleeding risk may be increased in patients on haemodialysis.[21] Another score used to assess bleeding risk on anticoagulation, specifically Warfarin or Coumadin, is the ATRIA score, which uses a weighted additive scale of clinical findings to determine bleeding risk stratification.[22]
The risks of bleeding are increased further when warfarin is combined with antiplatelet drugs such as clopidogrel, aspirin, or nonsteroidal anti-inflammatory drugs.[23]
Warfarin necrosis
[edit]A rare but serious complication resulting from treatment with warfarin is warfarin necrosis, which occurs more frequently shortly after commencing treatment in patients with a deficiency of protein C. Protein C is an innate anticoagulant that, like the procoagulant factors that warfarin inhibits, requires vitamin K-dependent carboxylation for its activity. Since warfarin initially decreases protein C levels faster than the coagulation factors, it can paradoxically increase the blood's tendency to coagulate when treatment is first begun (many patients when starting on warfarin are given heparin in parallel to combat this), leading to massive thrombosis with skin necrosis and gangrene of limbs. Its natural counterpart, purpura fulminans, occurs in children who are homozygous for certain protein C mutations.[24]
Osteoporosis
[edit]After initial reports that warfarin could reduce bone mineral density, several studies have demonstrated a link between warfarin use and osteoporosis-related fracture. A 1999 study in 572 women taking warfarin for deep venous thrombosis, risk of vertebral fracture and rib fracture was increased; other fracture types did not occur more commonly.[25] A 2002 study looking at a randomly selected selection of 1523 patients with osteoporotic fracture found no increased exposure to anticoagulants compared to controls, and neither did stratification of the duration of anticoagulation reveal a trend towards fracture.[26]
A 2006 retrospective study of 14,564 Medicare recipients showed that warfarin use for more than one year was linked with a 60% increased risk of osteoporosis-related fracture in men; there was no association in women. The mechanism was thought to be a combination of reduced intake of vitamin K, which is necessary for bone health, and inhibition by warfarin of vitamin K-mediated carboxylation of certain bone proteins, rendering them nonfunctional.[27]
Purple toe syndrome
[edit]Another rare complication that may occur early during warfarin treatment (usually within 3 to 8 weeks of commencement) is purple toe syndrome. This condition is thought to result from small deposits of cholesterol breaking loose and causing embolisms in blood vessels in the skin of the feet, which causes a blueish purple colour and may be painful.
It is typically thought to affect the big toe, but it affects other parts of the feet as well, including the bottom of the foot (plantar surface). The occurrence of purple toe syndrome may require discontinuation of warfarin.[28]
Calcification of valves and arteries
[edit]Several epidemiological studies have also implicated warfarin use in valvular and vascular calcification. No specific treatment is available, but some modalities are under investigation.[29]
Overdose
[edit]The major side effect of warfarin use is bleeding. Risk of bleeding is increased if the INR is out of range (due to accidental or deliberate overdose or due to interactions).[18] Many drug interactions can increase the effect of warfarin, also causing an overdose.[1]
For people who need rapid reversal of warfarin and have serious bleeding or who are having emergency surgery, the effects of warfarin can be reversed with vitamin K with prothrombin complex concentrate or fresh frozen plasma in addition to intravenous vitamin K.[30] Blood products should not be routinely used to reverse warfarin when vitamin K could work alone.[30]
Details on reversing warfarin are provided in clinical practice guidelines from the American College of Chest Physicians.[2] For patients with an international normalized ratio (INR) between 4.5 and 10.0, a small dose (about 1000 mcg = one milligram) of oral vitamin K is sufficient. When warfarin is being given and INR is in therapeutic range, simple discontinuation of the drug for five days is usually enough to reverse the effect and cause INR to drop below 1.5.[31]
Interactions
[edit]Warfarin interacts with many commonly used drugs, and the metabolism of warfarin varies greatly between patients.[1] Some foods have also been reported to interact with warfarin.[1] Apart from the metabolic interactions, highly protein bound drugs can displace warfarin from serum albumin and cause an increase in the INR.[32] This makes finding the correct dosage difficult, and accentuates the need of monitoring; when initiating a medication that is known to interact with warfarin (e.g. simvastatin), INR checks are increased or dosages adjusted until a new ideal dosage is found.
Many commonly used antibiotics, such as metronidazole or the macrolides, will greatly increase the effect of warfarin by reducing the metabolism of warfarin in the body. Other broad-spectrum antibiotics can reduce the amount of the normal bacterial flora in the bowel, which make significant quantities of vitamin K1, thus potentiating the effect of warfarin.[33] In addition, food that contains large quantities of vitamin K1 will reduce the warfarin effect.[1][10] Thyroid activity also appears to influence warfarin dosing requirements;[34] hypothyroidism (decreased thyroid function) makes people less responsive to warfarin treatment,[35] while hyperthyroidism (overactive thyroid) boosts the anticoagulant effect.[36] Several mechanisms have been proposed for this effect, including changes in the rate of breakdown of clotting factors and changes in the metabolism of warfarin.[34][37]
Excessive use of alcohol is also known to affect the metabolism of warfarin and can elevate the INR and thus increase the risk of bleeding.[38] The U.S. Food and Drug Administration (FDA) product insert on warfarin states that alcohol should be avoided.[39]
Warfarin also interacts with many herbs and spices,[40] some used in food (such as ginger and garlic) and others used purely for medicinal purposes (such as ginseng and Ginkgo biloba). All may increase bleeding and bruising in people taking warfarin; similar effects have been reported with borage (starflower) oil or fish oils.[41] St. John's Wort, sometimes recommended to help with mild to moderate depression, reduces the effectiveness of a given dose of warfarin; it induces the enzymes that break down warfarin in the body, causing a reduced anticoagulant effect.[42]
Between 2003 and 2004, the UK Committee on Safety of Medicines received several reports of increased INR and risk of haemorrhage in people taking warfarin and cranberry juice.[43][44][45] Data establishing a causal relationship is still lacking, and a 2006 review found no cases of this interaction reported to the FDA;[45] nevertheless, several authors have recommended that both doctors and patients be made aware of its possibility.[46] The mechanism behind the interaction is still unclear.[45]
Chemistry
[edit]Chemical synthesis
[edit]The succinct synthesis of warfarin starts with condensation of ortho-hydroxyacetophenone (1–2) with ethyl carbonate to give the b-ketoester as the intermediate shown in the enol form. Attack of the phenoxide on the ester grouping leads to cyclization and formation of the coumarin. Conjugate addition of the anion from that product to methyl styryl ketone gives the corresponding Michael adduct and thus warfarin.[47]
Molecular structure
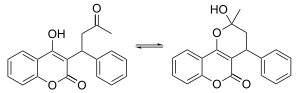
[edit]X-ray crystallographic studies of warfarin show that it exists in tautomeric form, as the cyclic hemiketal, which is formed from the 4-hydroxycoumadin and the ketone in the 3-position substituent.[48] However, the existence of many 4-hydroxycoumadin anticoagulants (for example phenprocoumon) that possess no ketone group in the 3-substituent to form such a structure, suggests that the hemiketal must hydrolyze to the 4-hydroxy form in order for warfarin to be active.[49]
Pharmacology
[edit]
Pharmacokinetics
[edit]Warfarin consists of a racemic mixture of two active enantiomers—R- and S- forms—each of which is cleared by different pathways. S-warfarin is 2-5 times more potent than the R-isomer in producing an anticoagulant response.[5]
Warfarin is slower-acting than the common anticoagulant heparin, though it has a number of advantages. Heparin must be given by injection, whereas warfarin is available orally. Warfarin has a long half-life and need only be given once a day. Heparin can also cause a prothrombotic condition, heparin-induced thrombocytopenia (an antibody-mediated decrease in platelet levels), which increases the risk for thrombosis. It takes several days for warfarin to reach the therapeutic effect since the circulating coagulation factors are not affected by the drug (thrombin has a half-life time of days). Warfarin's long half-life means that it remains effective for several days after it was stopped. Furthermore, if given initially without additional anticoagulant cover, it can increase thrombosis risk (see below). For these main reasons, hospitalised patients are usually given heparin with warfarin initially, the heparin covering the 3–5 day lag period and being withdrawn after a few days.
Mechanism of action
[edit]While warfarin is one of several drugs popularly referred to as a "blood thinner", this is a misnomer, since it does not affect the viscosity of blood.
Warfarin inhibits the vitamin K-dependent synthesis of biologically active forms of the calcium-dependent clotting factors II, VII, IX and X, as well as the regulatory factors protein C, protein S, and protein Z.[2][50] Other proteins not involved in blood clotting, such as osteocalcin, or matrix Gla protein, may also be affected.
The precursors of these factors require carboxylation of their glutamic acid residues to allow the coagulation factors to bind to phospholipid surfaces inside blood vessels, on the vascular endothelium. The enzyme that carries out the carboxylation of glutamic acid is gamma-glutamyl carboxylase. The carboxylation reaction will proceed only if the carboxylase enzyme is able to convert a reduced form of vitamin K (vitamin K hydroquinone) to vitamin K epoxide at the same time. The vitamin K epoxide is in turn recycled back to vitamin K and vitamin K hydroquinone by another enzyme, the vitamin K epoxide reductase (VKOR). Warfarin inhibits epoxide reductase[51] (specifically the VKORC1 subunit[52][53]), thereby diminishing available vitamin K and vitamin K hydroquinone in the tissues, which inhibits the carboxylation activity of the glutamyl carboxylase. When this occurs, the coagulation factors are no longer carboxylated at certain glutamic acid residues, and are incapable of binding to the endothelial surface of blood vessels, and are thus biologically inactive. As the body's stores of previously produced active factors degrade (over several days) and are replaced by inactive factors, the anticoagulation effect becomes apparent. The coagulation factors are produced, but have decreased functionality due to undercarboxylation; they are collectively referred to as PIVKAs (proteins induced [by] vitamin K absence/antagonism), and individual coagulation factors as PIVKA-number (e.g.PIVKA-II). The end result of warfarin use, therefore, is to diminish blood clotting in the patient.
When warfarin is newly started, it may promote clot formation temporarily. This is because the level of protein C and protein S are also dependent on vitamin K activity. Warfarin causes decline in protein C levels in first 36 hours. In addition, reduced levels of protein S lead to a reduction in activity of protein C (for which it is the co-factor) and therefore reduced degradation of factor Va and factor VIIIa. Although loading doses of warfarin over 5 mg also produce a precipitous decline in factor VII, resulting in an initial prolongation of the INR, full antithrombotic effect does not take place until significant reduction in factor II occurs days later. The haemostasis system becomes temporarily biased towards thrombus formation, leading to a prothrombotic state. Thus, when warfarin is loaded rapidly at greater than 5 mg per day, it is beneficial to co-administer heparin, an anticoagulant that acts upon antithrombin and helps reduce the risk of thrombosis, with warfarin therapy for four to five days, in order to have the benefit of anticoagulation from heparin until the full effect of warfarin has been achieved.[54][55]
Pharmacogenomics
[edit]Warfarin activity is determined partially by genetic factors. Polymorphisms in two genes (VKORC1 and CYP2C9) play a particularly large role in response to warfarin.
VKORC1 polymorphisms explain 30% of the dose variation between patients:[56] particular mutations make VKORC1 less susceptible to suppression by warfarin.[53] There are two main haplotypes that explain 25% of variation: low-dose haplotype group (A) and a high-dose haplotype group (B).[57] VKORC1 polymorphisms explain why African Americans are on average relatively resistant to warfarin (higher proportion of group B haplotypes), while Asian Americans are generally more sensitive (higher proportion of group A haplotypes).[57] Group A VKORC1 polymorphisms lead to a more rapid achievement of a therapeutic INR, but also a shorter time to reach an INR over 4, which is associated with bleeding.[58]
CYP2C9 polymorphisms explain 10% of the dose variation between patients,[56] mainly among Caucasian patients as these variants are rare in African American and most Asian populations.[59] These CYP2C9 polymorphisms do not influence time to effective INR as opposed toVKORC1, but does shorten the time to INR >4.[58]
Despite the promise of pharmacogenomic testing in warfarin dosing, its use in clinical practice is controversial. In August 2009 the Centers for Medicare and Medicaid Services concluded that "the available evidence does not demonstrate that pharmacogenomic testing of CYP2C9 or VKORC1 alleles to predict warfarin responsiveness improves health outcomes in Medicare beneficiaries."[60] A 2014 meta-analysis showed that using genotype-based dosing did not confer benefit in terms of time within therapeutic range, excessive anticoagulation (as defined by INR greater than 4), or a reduction in either major bleeding or thromboembolic events.[61]
History
[edit]In the early 1920s, there was an outbreak of a previously unrecognised cattle disease in the northern United States and Canada. Cattle were haemorrhaging after minor procedures, and on some occasions, spontaneously. For example, 21 out of 22 cows died after dehorning, and 12 out of 25 bulls died after castration. All of these animals had bled to death.[62]
In 1921, Frank Schofield, a Canadian veterinary pathologist, determined that the cattle were ingesting moldy silage made from sweet clover that functioned as a potent anticoagulant. Only spoiled hay made from sweet clover (grown in northern states of the USA and in Canada since the turn of the century) produced the disease.[63] Schofield separated good clover stalks and damaged clover stalks from the same hay mow, and fed each to a different rabbit. The rabbit that had ingested the good stalks remained well, but the rabbit that had ingested the damaged stalks died from a haemorrhagic illness. A duplicate experiment with a different sample of clover hay produced the same result.[62] In 1929, North Dakota veterinarian Dr L.M. Roderick demonstrated that the condition was due to a lack of functioning prothrombin.[64]
The identity of the anticoagulant substance in spoiled sweet clover remained a mystery until 1940. In 1933 Karl Paul Link and his lab of chemists working at the University of Wisconsin set out to isolate and characterize the haemorrhagic agent from the spoiled hay. It took five years for Link's student Harold A. Campbell to recover 6 mg of crystalline anticoagulant. Next, Link's student Mark A. Stahmann took over the project and initiated a large-scale extraction, isolating 1.8 g of recrystallized anticoagulant in about 4 months. This was enough material for Stahmann and Charles F. Huebner to check their results against Campbell's and to thoroughly characterize the compound. Through degradation experiments they established that the anticoagulant was 3,3'-methylenebis-(4-hydroxycoumarin), which they later named dicoumarol. They confirmed their results by synthesizing dicoumarol and proving in 1940 that it was identical to the naturally occurring agent.[65]
Dicoumarol was a product of the plant molecule coumarin (not to be confused with Coumadin, a later tradename for warfarin). Coumarin is now known to be present in many plants, and produces the notably sweet smell of freshly cut grass or hay and plants like sweet grass; in fact, the plant's high content of coumarin is responsible for the original common name of "sweet clover", which is named for its sweet smell, not its bitter taste.[62] They are present notably in woodruff (Galium odoratum, Rubiaceae), and at lower levels in licorice, lavender, and various other species. However, coumarins themselves do not influence clotting or warfarin-like action, but must first be metabolized by various fungi into compounds such as 4-hydroxycoumarin, then further (in the presence of naturally occurring formaldehyde) into dicoumarol, in order to have any anticoagulant properties. Fungal attack of the damaged and dying clover stalks explained the presence of the anticoagulant only in spoiled clover silages; dicoumarol is considered to be a fermentation product and mycotoxin.[66]
Over the next few years, numerous similar chemicals (specifically 4-hydroxycoumarins with a large aromatic substitutent at the 3 position) were found to have the same anticoagulant properties. The first drug in the class to be widely commercialized was dicoumarol itself, patented in 1941 and later used as a pharmaceutical. Karl Link continued working on developing more potent coumarin-based anticoagulants for use as rodent poisons, resulting in warfarin in 1948. The name "warfarin" stems from the acronym WARF, for Wisconsin Alumni Research Foundation + the ending -arin indicating its link with coumarin. Warfarin was first registered for use as a rodenticide in the US in 1948, and was immediately popular. Although warfarin was developed by Link, the Wisconsin Alumni Research Foundation financially supported the research and was assigned the patent.[67]
After an incident in 1951, where a US Army inductee unsuccessfully attempted suicide with multiple doses of warfarin in rodenticide and recovered fully after presenting to a hospital, and being treated with vitamin K (by then known as a specific antidote),[67] studies began in the use of warfarin as a therapeutic anticoagulant. It was found to be generally superior to dicoumarol, and in 1954 was approved for medical use in humans. An early recipient of warfarin was US president Dwight Eisenhower, who was prescribed the drug after having a heart attack in 1955.[67]
The exact mechanism of action remained unknown until it was demonstrated, in 1978, that warfarin inhibits the enzyme epoxide reductase and hence interferes with vitamin K metabolism.[51]
It has been theorized that Lavrenty Beria, Nikita Khrushchev and others conspired to use warfarin to poison the Soviet leader Joseph Stalin. Warfarin is tasteless and colourless, and produces symptoms similar to those that Stalin exhibited.[68]
Use as a pesticide
[edit]
To this day, the so-called "coumarins" (4-hydroxycoumarin derivatives) are used as rodenticides for controlling rats and mice in residential, industrial, and agricultural areas. Warfarin is both odorless and tasteless, and is effective when mixed with food bait, because the rodents will return to the bait and continue to feed over a period of days until a lethal dose is accumulated (considered to be 1 mg/kg/day over about six days). It may also be mixed with talc and used as a tracking powder, which accumulates on the animal's skin and fur, and is subsequently consumed during grooming. The LD50 is 50–500 mg/kg. The IDLH value is 100 mg/m³ (warfarin; various species).[69]
The use of warfarin itself as a rat poison is now declining, because many rat populations have developed resistance to it, and poisons of considerably greater potency are now available. Other 4-hydroxycoumarins used as rodenticides include coumatetralyl and brodifacoum, which is sometimes referred to as "super-warfarin", because it is more potent, longer-acting, and effective even in rat and mouse populations that are resistant to warfarin. Unlike warfarin, which is readily excreted, newer anticoagulant poisons also accumulate in the liver and kidneys after ingestion.[70]
Summary of significant drug interactions
[edit]The following interactions with other drugs are designated as significant:
Summary of significant drug interactions
[edit]The following interactions with other drugs are designated as significant:
References
[edit]- ^ a b c d e f Holbrook AM, Pereira JA, Labiris R, McDonald H, Douketis JD, Crowther M, Wells PS (May 2005). "Systematic overview of warfarin and its drug and food interactions". Arch. Intern. Med. 165 (10): 1095–106. doi:10.1001/archinte.165.10.1095. PMID 15911722.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b c d Ansell J, Hirsh J, Hylek E; et al. (2008). "Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians evidence-based clinical practice guidelines (8th Edition)". Chest. 133 (6 Suppl): 160S–198S. doi:10.1378/chest.08-0670. PMID 18574265.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Holford NH. Clinical pharmacokinetics and pharmacodynamics of warfarin. Understanding the dose-effect relationship. Clin Pharmacokinet 1986; 11: 483–504.
- ^ "coumadin". The American Society of Health-System Pharmacists. Retrieved 3 April 2011.
- ^ a b c Hirsh J, Fuster V, Ansell J, Halperin JL (2003). "American Heart Association/American College of Cardiology Foundation guide to warfarin therapy". J. Am. Coll. Cardiol. 41 (9): 1633–52. doi:10.1016/S0735-1097(03)00416-9. PMID 12742309.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Hirsh J, O'Donnell M, Eikelboom JW (July 2007). "Beyond unfractionated heparin and warfarin: current and future advances". Circulation. 116 (5): 552–60. doi:10.1161/CIRCULATIONAHA.106.685974. PMID 17664384. S2CID 8596503.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Perry, DJ (2010). "Point-of-care testing in haemostasis". Br J Haematol. 150 (5): 501–14. doi:10.1111/j.1365-2141.2010.08223.x. PMID 20618331. S2CID 32069018.
- ^ Baglin TP, Keeling DM, Watson HG (February 2006). "Guidelines on oral anticoagulation (warfarin): third edition—2005 update". Br. J. Haematol. 132 (3): 277–85. doi:10.1111/j.1365-2141.2005.05856.x. PMID 16409292. S2CID 9263819.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ National Institutes of Health. "important information to know when you are taking : Coumadine and vitamine K" (PDF). Retrieved 27 March 2014.
- ^ a b c d Holbrook; et al. (2012). "Evidence-based management of anticoagulant therapy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines". Chest. 141 (2 Suppl): e152S–84S. doi:10.1378/chest.11-2295. PMC 3278055. PMID 22315259.
{{cite journal}}: Explicit use of et al. in:|last=(help) - ^ "Point-of-Care Guides: May 15, 2005. American Family Physician". Archived from the original on 29 September 2007. Retrieved 20 August 2007.
- ^ "Warfarin diet: What foods should I avoid?". Mayoclinic.com. Retrieved 9 August 2011.
- ^ a b Ansell J, Jacobson A, Levy J, Völler H, Hasenkam JM (March 2005). "Guidelines for implementation of patient self-testing and patient self-management of oral anticoagulation. International consensus guidelines prepared by International Self-Monitoring Association for Oral Anticoagulation" (PDF). Int. J. Cardiol. 99 (1): 37–45. doi:10.1016/j.ijcard.2003.11.008. PMID 15721497.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Heneghan C, Alonso-Coello P, Garcia-Alamino JM, Perera R, Meats E, Glasziou P (February 2006). "Self-monitoring of oral anticoagulation: a systematic review and meta-analysis" (PDF). Lancet. 367 (9508): 404–11. doi:10.1016/S0140-6736(06)68139-7. PMID 16458764. S2CID 1494933.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b c d e Macina, Orest T.; Schardein, James L. (2007). "Warfarin". Human Developmental Toxicants. Boca Raton: CRC Taylor & Francis. pp. 193–4. ISBN 978-0-8493-7229-2.
{{cite book}}: CS1 maint: multiple names: authors list (link) Retrieved on 15 December 2008 through Google Book Search. - ^ a b c d e Loftus, Christopher M. (1995). "Fetal toxicity of common neurosurgical drugs". Neurosurgical Aspects of Pregnancy. Park Ridge, Ill: American Association of Neurological Surgeons. pp. 11–3. ISBN 1-879284-36-7.
- ^ a b Bates, SM (2012). "VTE, thrombophilia, antithrombotic therapy, and pregnancy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines". Chest. 141 (2 Suppl): e691S–736S. doi:10.1378/chest.11-2300. PMC 3278054. PMID 22315276.
- ^ a b Garcia D, Crowther MA, Ageno W (2010). "Practical management of coagulopathy associated with warfarin". BMJ. 19 (340): c1813. doi:10.1136/bmj.c1813. PMID 20404060. S2CID 37076001.
{{cite journal}}: CS1 maint: multiple names: authors list (link) Cite error: The named reference "Garcia2010" was defined multiple times with different content (see the help page). - ^ Camm AJ, Lip GY, De Caterina R, Savelieva I, Atar D, Hohnloser SH, Hindricks G, Kirchhof P; ESC Committee for Practice Guidelines (CPG) (2012). "2012 focused update of the ESC Guidelines for the management of atrial fibrillation: an update of the 2010 ESC Guidelines for the management of atrial fibrillation. Developed with the special contribution of the European Heart Rhythm Association". Eur Heart J. 33 (21): 2719–47. doi:10.1093/eurheartj/ehs253. PMID 22922413.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Shoeb M, Fang MC (2013). "Assessing bleeding risk in patients taking anticoagulants". J Thromb Thrombolysis. 35 (3): 312–9. doi:10.1007/s11239-013-0899-7. PMC 3888359. PMID 23479259.
- ^ Elliott MJ, Zimmerman D, Holden RM (2007). "Warfarin anticoagulation in hemodialysis patients: a systematic review of bleeding rates". Am. J. Kidney Dis. 50 (3): 433–40. doi:10.1053/j.ajkd.2007.06.017. PMID 17720522.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Fang MC; et al. (2011). "A New Risk Scheme to Predict Warfarin-Associated Hemorrhage: The ATRIA (Anticoagulation and Risk Factors in Atrial Firbrillation)". J Am Coll Cardiol. 58 (4): 395–401. doi:10.1016/j.jacc.2011.03.031. PMC 3175766. PMID 21757117.
{{cite journal}}: Explicit use of et al. in:|author=(help) - ^ Delaney JA, Opatrny L, Brophy JM, Suissa S (2007). "Drug drug interactions between antithrombotic medications and the risk of gastrointestinal bleeding". CMAJ. 177 (4): 347–51. doi:10.1503/cmaj.070186. PMC 1942107. PMID 17698822.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Chan YC, Valenti D, Mansfield AO, Stansby G (2000). "Warfarin induced skin necrosis". Br J Surg. 87 (3): 266–72. doi:10.1046/j.1365-2168.2000.01352.x. PMID 10718793. S2CID 30687579.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Caraballo PJ, Heit JA, Atkinson EJ, Silverstein MD, O'Fallon WM, Castro MR, Melton LJ (1999). "Long-term use of oral anticoagulants and the risk of fracture". Arch. Intern. Med. 159 (15): 1750–6. doi:10.1001/archinte.159.15.1750. PMID 10448778.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Pilon D, Castilloux AM, Dorais M, LeLorier J (2004). "Oral anticoagulants and the risk of osteoporotic fractures among elderly". Pharmacoepidemiol Drug Saf. 13 (5): 289–94. doi:10.1002/pds.888. PMID 15133779. S2CID 45496277.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Gage BF, Birman-Deych E, Radford MJ, Nilasena DS, Binder EF (2006). "Risk of osteoporotic fracture in elderly patients taking warfarin: results from the National Registry of Atrial Fibrillation 2". Arch. Intern. Med. 166 (2): 241–6. doi:10.1001/archinte.166.2.241. PMID 16432096.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Talmadge DB, Spyropoulos AC (2003). "Purple toes syndrome associated with warfarin therapy in a patient with antiphospholipid syndrome". Pharmacotherapy. 23 (5): 674–7. doi:10.1592/phco.23.5.674.32200. PMID 12741443. S2CID 28632135.
- ^ Palaniswamy C, Sekhri A, Aronow WS, Kalra A, Peterson SJ (February 2011). "Association of warfarin use with valvular and vascular calcification: a review". Clin Cardiol. 34 (2): 74–81. doi:10.1002/clc.20865. PMC 6652734. PMID 21298649.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b American Association of Blood Banks (24 April 2014), "Five Things Physicians and Patients Should Question", Choosing Wisely: an initiative of the ABIM Foundation, American Association of Blood Banks, retrieved 25 July 2014
- ^ Crowther MA, Douketis JD, Schnurr T, Steidl L, Mera V, Ultori C, Venco A, Ageno W (August 2002). "Oral vitamin K lowers the international normalized ratio more rapidly than subcutaneous vitamin K in the treatment of warfarin-associated coagulopathy. A randomized, controlled trial". Ann. Intern. Med. 137 (4): 251–4. doi:10.7326/0003-4819-137-4-200208200-00009. PMID 12186515. S2CID 10450603.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Gage BF, Fihn SD, White RH (October 2000). "Management and dosing of warfarin therapy". Am. J. Med. 109 (6): 481–8. doi:10.1016/S0002-9343(00)00545-3. PMID 11042238.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Juurlink DN (August 2007). "Drug interactions with warfarin: what clinicians need to know". CMAJ. 177 (4): 369–71. doi:10.1503/cmaj.070946. PMC 1942100. PMID 17698826.
- ^ a b Kurnik D, Loebstein R, Farfel Z, Ezra D, Halkin H, Olchovsky D (March 2004). "Complex drug-drug-disease interactions between amiodarone, warfarin, and the thyroid gland". Medicine (Baltimore). 83 (2): 107–13. doi:10.1097/01.md.0000123095.65294.34. PMID 15028964. S2CID 43173080.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Stephens MA, Self TH, Lancaster D, Nash T (December 1989). "Hypothyroidism: effect on warfarin anticoagulation". South Med J. 82 (12): 1585–6. doi:10.1097/00007611-198912000-00035. PMID 2595433.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Chute JP, Ryan CP, Sladek G, Shakir KM; Ryan; Sladek; Shakir (1997). "Exacerbation of warfarin-induced anticoagulation by hyperthyroidism". Endocr Pract. 3 (2): 77–9. doi:10.4158/EP.3.2.77. PMID 15251480.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Kellett HA, Sawers JS, Boulton FE, Cholerton S, Park BK, Toft AD (January 1986). "Problems of anticoagulation with warfarin in hyperthyroidism". Q J Med. 58 (225): 43–51. PMID 3704105.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Weathermon R, Crabb DW (1999). "Alcohol and medication interactions". Alcohol Res Health. 23 (1): 40–54. PMC 6761694. PMID 10890797.
- ^ Cite error: The named reference
TGAwas invoked but never defined (see the help page). - ^ Austin, Steve and Batz, Forrest (1999). Lininger, Schuyler W. (ed.). A-Z guide to drug-herb-vitamin interactions: how to improve your health and avoid problems when using common medications and natural supplements together. Roseville, Calif: Prima Health. p. 224. ISBN 0-7615-1599-2.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ "Information Pharmacists' news" (PDF). Information Centre Bulletin. 1 Lambeth High Street, London SE1 7JN: Royal Pharmaceutical Society of Great Britain. February 2008. p. 1. Retrieved 14 January 2009.
{{cite web}}: CS1 maint: location (link) [dead link] - ^ Dr Jo Barnes BPharm MRPharmS; Working Group on Complementary Medicine (September 2002). "Herb-medicine interactions: St John's Wort (Hypericum perforatum) Useful information for pharmacist" (PDF). 1 Lambeth High Street, London SE1 7JN: Royal Pharmaceutical Society of Great Britain. p. 5. Retrieved 14 January 2009.
{{cite web}}: CS1 maint: location (link) - ^ "Cranberry juice clot drug warning". BBC news. 18 September 2003. Retrieved 18 May 2008.
- ^ Suvarna R, Pirmohamed M, Henderson L (December 2003). "Possible interaction between warfarin and cranberry juice". BMJ. 327 (7429): 1454. doi:10.1136/bmj.327.7429.1454. PMC 300803. PMID 14684645.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b c Aston JL, Lodolce AE, Shapiro NL (September 2006). "Interaction between warfarin and cranberry juice". Pharmacotherapy. 26 (9): 1314–9. doi:10.1592/phco.26.9.1314. PMID 16945054. S2CID 28468365.
{{cite journal}}: CS1 maint: multiple names: authors list (link)Free full text with registration at Medscape - ^ Pham DQ, Pham AQ (March 2007). "Interaction potential between cranberry juice and warfarin". Am J Health Syst Pharm. 64 (5): 490–4. doi:10.2146/ajhp060370. PMID 17322161.
- ^ Blicke, F. F.; Swisher, R. D. (1934). "The Interaction of the Acid Chloride of 2-Benzoylbenzoic Acid with Phenols. I. Aryl Esters and Phenylaryloxyphthalides1". Journal of the American Chemical Society. 56 (4): 902–904. doi:10.1021/ja01319a042.
- ^ Valente, E. J.; Trager, W. F.; Jensen, L. H. (1975). "The crystal and molecular structure and absolute configuration of (−)-(S)-warfarin". Acta Cryst. B. 31 (4): 954–960. doi:10.1107/S056774087500427X.
- ^ Karlsson BC, Rosengren AM, Andersson PO, Nicholls IA (September 2007). "The spectrophysics of warfarin: implications for protein binding". J Phys Chem B. 111 (35): 10520–8. doi:10.1021/jp072505i. PMID 17691835.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Freedman MD (March 1992). "Oral anticoagulants: pharmacodynamics, clinical indications and adverse effects". J Clin Pharmacol. 32 (3): 196–209. doi:10.1002/j.1552-4604.1992.tb03827.x. PMID 1564123. S2CID 38963632.
- ^ a b Whitlon DS, Sadowski JA, Suttie JW (1978). "Mechanism of coumarin action: significance of vitamin K epoxide reductase inhibition". Biochemistry. 17 (8): 1371–7. doi:10.1021/bi00601a003. PMID 646989.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Li T, Chang CY, Jin DY, Lin PJ, Khvorova A, Stafford DW (2004). "Identification of the gene for vitamin K epoxide reductase". Nature. 427 (6974): 541–4. doi:10.1038/nature02254. PMID 14765195. S2CID 4424554.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Rost S, Fregin A, Ivaskevicius V, Conzelmann E, Hörtnagel K, Pelz HJ, Lappegard K, Seifried E, Scharrer I, Tuddenham EG, Müller CR, Strom TM, Oldenburg J (February 2004). "Mutations in VKORC1 cause warfarin resistance and multiple coagulation factor deficiency type 2". Nature. 427 (6974): 537–41. doi:10.1038/nature02214. PMID 14765194. S2CID 4424197.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Litin SC, Gastineau DA (March 1995). "Current concepts in anticoagulant therapy". Mayo Clin. Proc. 70 (3): 266–72. doi:10.4065/70.3.266. PMID 7861815.
- ^ Wittkowsky AK (2005). "Why warfarin and heparin need to overlap when treating acute venous thromboembolism". Dis Mon. 51 (2–3): 112–5. doi:10.1016/j.disamonth.2005.03.005. PMID 15900262.
- ^ a b Wadelius M, Chen LY, Downes K, Ghori J, Hunt S, Eriksson N, Wallerman O, Melhus H, Wadelius C, Bentley D, Deloukas P (2005). "Common VKORC1 and GGCX polymorphisms associated with warfarin dose". Pharmacogenomics J. 5 (4): 262–70. doi:10.1038/sj.tpj.6500313. PMID 15883587. S2CID 32845262.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Rieder MJ, Reiner AP, Gage BF, Nickerson DA, Eby CS, McLeod HL, Blough DK, Thummel KE, Veenstra DL, Rettie AE (June 2005). "Effect of VKORC1 haplotypes on transcriptional regulation and warfarin dose". N. Engl. J. Med. 352 (22): 2285–93. doi:10.1056/NEJMoa044503. PMID 15930419.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Schwarz UI, Ritchie MD, Bradford Y, Li C, Dudek SM, Frye-Anderson A, Kim RB, Roden DM, Stein CM (March 2008). "Genetic determinants of response to warfarin during initial anticoagulation". N. Engl. J. Med. 358 (10): 999–1008. doi:10.1056/NEJMoa0708078. PMC 3894627. PMID 18322281.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Sanderson S, Emery J, Higgins J (2005). "CYP2C9 gene variants, drug dose, and bleeding risk in warfarin-treated patients: a HuGEnet systematic review and meta-analysis". Genet. Med. 7 (2): 97–104. doi:10.1002/ajmg.a.30391. PMID 15714076. S2CID 34195357.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Jensen TS, Jacques LB, Ciccanti M, Long K, Eggleston L, Roche J (3 August 2009). "Decision Memo for Pharmacogenomic Testing for Warfarin Response (CAG-00400N)". Centers for Medicare and Medicaid Services. Retrieved 27 March 2011.
{{cite web}}: CS1 maint: multiple names: authors list (link) - ^ Stergiopoulos K, Brown DL (August 2014). "Genotype-Guided vs Clinical Dosing of Warfarin and Its Analogues: Meta-analysis of Randomized Clinical Trials". JAMA Intern Med. 174 (8): 1330–1338. doi:10.1001/jamainternmed.2014.2368. PMID 24935087.
- ^ a b c Laurence, D.R. (1973). Clinical Pharmacology. Peter Kneebone. Edinburgh, London and New York: Churchill Livingstone. pp. 23.4–23.5. ISBN 0-443-04990-4.
- ^ Schofield FW (1924). "Damaged sweet clover; the cause of a new disease in cattle simulating haemorrhagic septicemia and blackleg". J Am Vet Med Assoc. 64: 553–6.
- ^ Roderick LM (1931). "A problem in the coagulation of the blood; "sweet clover disease of the cattle"". Am J Physiol. 96: 413–6. doi:10.1152/ajplegacy.1931.96.2.413.PDF (subscriber only).
- ^ Stahmann MA, Huebner CF, Link KP (1 April 1941). "Studies on the hemorrhagic sweet clover disease. V. Identification and synthesis of the hemorrhagic agent". J Biol Chem. 138 (2): 513–27. doi:10.1016/S0021-9258(18)51377-6.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Bye, A., King, H. K., 1970. The biosynthesis of 4-hydroxycoumarin and dicoumarol by Aspergillus fumigatus Fresenius. Biochemical Journal 117, 237–245.
- ^ a b c Link KP (1 January 1959). "The discovery of dicumarol and its sequels". Circulation. 19 (1): 97–107. doi:10.1161/01.CIR.19.1.97. PMID 13619027. S2CID 21634182.
- ^ Naumov, Vladimir Pavlovich; Brent, Jonathan (2003). Stalin's last crime: the plot against the Jewish doctors, 1948–1953. London: HarperCollins. ISBN 0-06-019524-X.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ United States Occupational Safety and Health Administration (OSHA) (16 August 1996). "Documentation for Immediately Dangerous To Life or Health Concentrations (IDLHs): Warfarin". Centers for Disease Control and Prevention. Retrieved 7 July 2008.
- ^ Charles T. Eason; Mark Wickstrom (2001). "2. Anticoagulant poisons" (PDF). Vertebrate pesticide toxicology manual (poisons). New Zealand Department of Conservation. pp. 41–74. ISBN 0-478-22035-9. Retrieved 7 July 2008.
External links
[edit]- Historical information on warfarin from the Wisconsin Alumni Research Foundation
- Online sweet clover disease and warfarin historical review
- U.S. National Library of Medicine: Drug Information Portal–Warfarin
- Warfarin bound to proteins in the PDB: R-warfarin,S-warfarin
- CDC - NIOSH Pocket Guide to Chemical Hazards
- Pesticide Properties DataBase (PPDB)
Online Bleeding Risk Calculators
[edit]- ATRIA Bleeding Risk Score from MDCalc
- HAS-BLED Score from MDCalc