Rimcazole
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
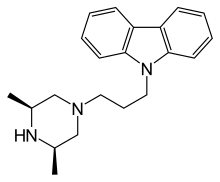
| Formula | C21H27N3 |
| Molar mass | 321.468 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Rimcazole is an antagonist[1] of the sigma receptor[2] as well as a dopamine reuptake inhibitor.[3] Sigma receptors are thought to be involved in the drug psychosis that can be induced by some drugs such as phencyclidine and cocaine, and rimcazole was originally researched as a potential antipsychotic with a different mechanism of action to traditional antipsychotic drugs. Trials proved inconclusive and rimcazole was not pursued for this application, but other sigma antagonists continue to be researched for a variety of potential applications.[4] Rimcazole has been shown to reduce the effects of cocaine,[5] and analogues of rimcazole have been shown to be highly effective at blocking the convulsions caused by cocaine overdose in animal models.[6] Isothiocyanate derivatives of rimcazole acting as irreversible dopamine transporter (DAT) blockers have been developed.[3]
See also
[edit]References
[edit]- ^ Gilmore DL, Liu Y, Matsumoto RR (2004). "Review of the pharmacological and clinical profile of rimcazole". CNS Drug Reviews. 10 (1): 1–22. doi:10.1111/j.1527-3458.2004.tb00001.x. PMC 6741722. PMID 14978511.
- ^ Eaton MJ, Lookingland KJ, Moore KE (September 1996). "The sigma ligand rimcazole activates noradrenergic neurons projecting to the paraventricular nucleus and increases corticosterone secretion in rats". Brain Research. 733 (2): 162–166. doi:10.1016/0006-8993(96)00290-9. PMID 8891298. S2CID 42885767.
- ^ a b Husbands SM, Izenwasser S, Loeloff RJ, Katz JL, Bowen WD, Vilner BJ, Newman AH (December 1997). "Isothiocyanate derivatives of 9-[3-(cis-3,5-dimethyl-1-piperazinyl)propyl]carbazole (rimcazole): irreversible ligands for the dopamine transporter". J Med Chem. 40 (26): 4340–46. doi:10.1021/jm9705519. PMID 9435903.
- ^ Volz HP, Stoll KD (November 2004). "Clinical trials with sigma ligands". Pharmacopsychiatry. 37 (Suppl 3): S214–S220. doi:10.1055/s-2004-832680. PMID 15547788. S2CID 260238757.
- ^ Katz JL, Libby TA, Kopajtic T, Husbands SM, Newman AH (May 2003). "Behavioral effects of rimcazole analogues alone and in combination with cocaine". European Journal of Pharmacology. 468 (2): 109–119. doi:10.1016/s0014-2999(03)01638-8. PMID 12742518.
- ^ Matsumoto RR, Hewett KL, Pouw B, Bowen WD, Husbands SM, Cao JJ, Newman AH (December 2001). "Rimcazole analogs attenuate the convulsive effects of cocaine: correlation with binding to sigma receptors rather than dopamine transporters". Neuropharmacology. 41 (7): 878–886. doi:10.1016/s0028-3908(01)00116-2. PMID 11684152. S2CID 44328858.
