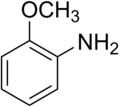
o-Anisidine
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-Methoxyaniline[1] | |||
| Other names
ortho-Anisidine
2-Anisidine ortho-Aminoanisole o-Methoxyaniline 2-Methoxy-1-aminobenzene 2-Methoxyphenylamine | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.001.785 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 2431 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties[2] | |||
| C7H9NO | |||
| Molar mass | 123.155 g·mol−1 | ||
| Appearance | Yellow liquid, turns brown upon exposure to air | ||
| Density | 1.0923 g/cm3 | ||
| Melting point | 6.2 °C (43.2 °F; 279.3 K) | ||
| Boiling point | 224 °C (435 °F; 497 K) | ||
| 1.5 g/100 ml | |||
| Solubility | soluble in ethanol, diethyl ether, acetone, benzene | ||
| -80.44·10−6 cm3/mol | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
potential occupational carcinogen[3] | ||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H301, H311, H331, H341, H350 | |||
| P201, P202, P261, P264, P270, P271, P280, P281, P301+P310, P302+P352, P304+P340, P308+P313, P311, P312, P321, P322, P330, P361, P363, P403+P233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 118 °C (244 °F; 391 K) (open cup) | ||
| 415 °C (779 °F; 688 K) | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
2000 mg/kg (rat, oral) 1400 mg/kg (mouse, oral) 870 mg/kg (rabbit, oral)[4] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 0.5 mg/m3 [skin][3] | ||
REL (Recommended)
|
: Ca TWA 0.5 mg/m3 [skin][3] | ||
IDLH (Immediate danger)
|
50 mg/m3[3] | ||
| Related compounds | |||
Related compounds
|
m-Anisidine p-Anisidine | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
o-Anisidine (2-anisidine) is an organic compound with the formula CH3OC6H4NH2. A colorless liquid, commercial samples can appear yellow owing to air oxidation. It is one of three isomers of the methoxy-containing aniline derivative.
Production and use
[edit]It is prepared via methanolysis of 2-chloronitrobenzene:[5]
- NaOCH3 + ClC6H4NO2 → CH3OC6H4NO2 + NaCl
The resulting o-nitroanisole is reduced to o-anisidine.
o-Anisidine is used in the manufacture of dyes. It is nitrated to give 4-nitroanisidine. It is also a precursor to o-dianisidine.
One special use is as a heartwood indicator. An acid solution of o-anisidine is diazotized by adding a sodium nitrite solution. This mixture is applied to the wood and by reaction with polyphenols in the heartwood a reddish brown azo dye is formed.

Direct Blue 15 is an azo dye produced from o-anisidine
Safety and environmental aspects
[edit]o-Anisidine is a dangerous pollutant from the production of dyes. It is listed as RCRA hazardous waste, with the code K181.[6] The International Agency for Research on Cancer (IARC) has classified o-anisidine as a Group 2B, possible human carcinogen.[7]
References
[edit]- ^ Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 669. doi:10.1039/9781849733069-00648. ISBN 978-0-85404-182-4.
The names 'toluidine', 'anisidine', and 'phenetidine' for which o-, m-, and p- have been used to distinguish isomers, and 'xylidine' for which numerical locants, such as 2,3-, have been used, are no longer recommended, nor are the corresponding prefixes 'toluidine', 'anisidino', 'phenetidine', and 'xylidino'.
- ^ Weast, Robert C., ed. (1981). CRC Handbook of Chemistry and Physics (62nd ed.). Boca Raton, Florida: CRC Press. p. C-98. ISBN 0-8493-0462-8.
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0034". National Institute for Occupational Safety and Health (NIOSH).
- ^ "o-Anisidine". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ Gerald Booth (2007). "Nitro Compounds, Aromatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a17_411. ISBN 978-3527306732.
- ^ "Hazardous Waste". 2015-07-23.
- ^ "o-Anisidine".
External links
[edit]- International Chemical Safety Card 0970
- NIOSH Pocket Guide to Chemical Hazards. "#0034". National Institute for Occupational Safety and Health (NIOSH).