Acyl-CoA:lysocardiolipin acyltransferase-1
| Acyl-CoA:lysocardiolipin acyltransferase-1 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | ALCAT1 | ||||||
| Alt. symbols | LCLAT1, LYCAT | ||||||
| NCBI gene | 253558 | ||||||
| HGNC | 26756 | ||||||
| OMIM | 614241 | ||||||
| RefSeq | NM_182551.3 | ||||||
| UniProt | Q6UWP7 | ||||||
| Other data | |||||||
| Locus | Chr. 2 p23.1 | ||||||
| |||||||
Acyl-CoA:lysocardiolipin acyltransferase-1 (ALCAT1) is a polyglycerophospholipid acyltransferase of the endoplasmic reticulum which is primarily known for catalyzing the acylation of monolysocardiolipin back into cardiolipin, although it does catalyze the acylation of other polyglycerophospholipids.[1]
Overall reaction:
- monolysocardiolipin(MLCL) + acyl-CoA = cardiolipin + CoA
ALCAT1 is widely distributed throughout the body, with the highest concentrations being in the heart and liver.[2]
Mechanism
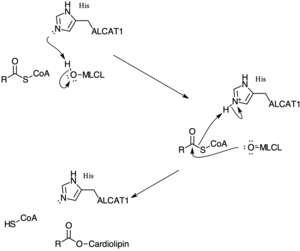
[edit]ALCAT1 shares similar mechanism with other acyltransferases that facilitates biosynthesis of esters from acyl-CoA and alcohol. In the first step, the free hydroxyl group on monolysocardiolipin is deprotonated to make a good nucleophilic attacker.[3]
Biological Function
[edit]A cardiolipin molecule achieves optimal biochemical functionality when all four fatty acyls it incorporates are linoleic.[4] This composition of fatty acyl is also known as tetralinoleoyl cardiolipin (TLCL). However, newly synthesized cardiolipin, also known as nascent cardiolipin, does not have such fatty acyl composition. Nascent cardiolipin has to go through a remodeling process, which involves deacylation to monolysocardiolipin (MLCL), then re-acylation back to cardiolipin (ideally TLCL).[5] When first discovered, ALCAT1 was believed to be a member of the group of enzymes that facilitates this important re-acylation of MLCL to TLCL.[2] However, after comparing its activity with other members within this group, including MLCLAT and Tafazzin (TAZ), new theories have been proposed to correlate ALCAT1 activity with oxidative stress and aging.[6]
Relation to Oxidative Stress and Aging
[edit]
While competing against MLCLAT and Tafazzin (TAZ) in re-acylation of MLCL back to cardiolipin, ALCAT1 has much worse selectivity in linoleic acyl.[7] ALCAT1 occasionally remodels cardiolipins with very-long-chain fatty acyls, such as docosahexaenoic (DHA) and arachidonic, leading to cardiolipins with not only non-optimal functionality compared to that of tetralinoleoyl cardiolipin (TLCL), but also vulnerability to oxidation. Subsequent mitochondrial dysfunction leads to more oxidative stress and reactive oxygen species (ROS), and consequently faster depletion of physiological cardiolipin due to oxidation. Moreover, the activity of ALCAT1 is up-regulated by oxidative stress, which closes the loop of a vicious cycle that is implicated in the pathogenesis of various age-related diseases.[6][8]
In animal studies, knockout of ALCAT1 improved cardiac function and inhibited the injury of the heart and kidney in mice with myocardial infraction. An increase of ALCAT1 level has been observed in skeletal muscles of denervation rats. Deficiency of ALCAT1 gene prevented mitochondrial fragmentation from oxidative stress and increased the thickness of the skeletal muscle fibers. Additionally, inhibition of ALCAT1 prevented SOD1 protein aggregation and attenuated skeletal muscle atrophy in SOD1 transgenic mice.[9]
Clinical Significance
[edit]Accumulating evidence suggests that over-expression of ALCAT1 is involved in pathological cardiolipin remodeling and mitochondrial bioenergetics. Up-regulated expression of ALCAT1 can increase the fraction of cardiolipins with aberrant acyl compositions including enrichment of cardiolipins with docosahexaenoic acyls, leading to tetralinoleoyl cardiolipins (TLCL) depletion. That renders cardiolipins high sensitivity to oxidative damage by reactive oxygen species (ROS), which has been linked to mitochondrial dysfunction associated with various metabolic, cardiovascular and neurodegenerative diseases, such as obesity, nonalcoholic fatty liver disease, coronary heart disease, and Parkinson's disease.[10] Therefore, ALCAT1 is suggested as a novel therapeutic target for the treatment of these diseases.[11]

References
[edit]- ^ Cao J, Shen W, Chang Z, Shi Y (April 2009). "ALCAT1 is a polyglycerophospholipid acyltransferase potently regulated by adenine nucleotide and thyroid status". American Journal of Physiology. Endocrinology and Metabolism. 296 (4): E647-53. doi:10.1152/ajpendo.90761.2008. PMC 3734525. PMID 19106248.
- ^ a b Cao J, Liu Y, Lockwood J, Burn P, Shi Y (July 2004). "A novel cardiolipin-remodeling pathway revealed by a gene encoding an endoplasmic reticulum-associated acyl-CoA:lysocardiolipin acyltransferase (ALCAT1) in mouse". The Journal of Biological Chemistry. 279 (30): 31727–34. doi:10.1074/jbc.M402930200. PMID 15152008.
- ^ Röttig, Annika (June 2013). "Acyltransferases in Bacteria". Microbiology and Molecular Biology Reviews. 77 (2): 277–321. doi:10.1128/MMBR.00010-13. PMC 3668668. PMID 23699259.
- ^ Schlame M, Horvath L, Vigh L (1994). "Relationship between lipid saturation and lipid-protein interaction in liver mitochondria modified by catalytic hydrogenation with reference to cardiolipin molecular species". Biochem. J. 265 (1): 79–85. doi:10.1042/bj2650079. PMC 1136616. PMID 2154183.
- ^ Duncan, Anna L. (June 30, 2020). "Monolysocardiolipin (MLCL) interactions with mitochondrial membrane proteins". Biochem Soc Trans. 48 (3): 993–1004. doi:10.1042/BST20190932. PMC 7329354. PMID 32453413.
- ^ a b c d Zhang, Jun; Shi, Yuguang (2022-06-12). "In Search of the Holy Grail: Toward a Unified Hypothesis on Mitochondrial Dysfunction in Age-Related Diseases". Cells. 11 (12): 1906. doi:10.3390/cells11121906. ISSN 2073-4409. PMC 9221202. PMID 35741033.
- ^ Taylor, W. A. (2009). "Identification of the human mitochondrial linoleoyl-coenzyme A monolysocardiolipin acyltransferase (MLCL AT-1)". J. Biol. Chem. 284 (44): 30360–30371. doi:10.1074/jbc.M109.048322. PMC 2781591. PMID 19737925.
- ^ Liu, X. (2012). "Ablation of ALCAT1 mitigates hypertrophic cardiomyopathy through effects on oxidative stress and mitophagy". Mol. Cell. Biol. 32 (21): 4493–4504. doi:10.1128/MCB.01092-12. PMC 3486149. PMID 22949503.
- ^ Ren, Wujing; Xu, Zujie; Pan, Shou; Ma, Yixuan; Li, Hangzhuo; Wu, Fangnan; Bo, Wenyan; Cai, Mengxin; Tian, Zhenjun (2022-11-20). "Irisin and ALCAT1 mediated aerobic exercise-alleviated oxidative stress and apoptosis in skeletal muscle of mice with myocardial infarction". Free Radical Biology & Medicine. 193 (Pt 2): 526–537. doi:10.1016/j.freeradbiomed.2022.10.321. ISSN 1873-4596. PMID 36336228. S2CID 253329656.
- ^ Song, C. (2019). "Cardiolipin remodeling by ALCAT1 links mitochondrial dysfunction to Parkinson's diseases". Aging Cell. 18 (3): e12941. doi:10.1111/acel.12941. PMC 6516155. PMID 30838774.
- ^ Huang, Hui; Zhou, Renmei; Wen, He; Xu, Zhixiang; Zhang, Shengmei; Wang, Weixue; Krosky, Daniel; Miller, Robyn (2022-09-15). "Synthesis of biotinylated-LPG as a chemical biology tool enabling discovery of ALCAT1 modulators". Bioorganic & Medicinal Chemistry Letters. 72: 128861. doi:10.1016/j.bmcl.2022.128861. ISSN 1464-3405. PMID 35718103. S2CID 249796710.
