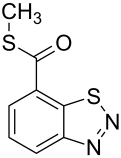
Acibenzolar-S-methyl
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
S-Methyl 1,2,3-benzothiadiazole-7-carbothioate | |||
| Other names
BTH, CGA245704
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.101.876 | ||
| EC Number |
| ||
| MeSH | S-methyl+benzo(1,2,3)thiadiazole-7-carbothioate | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties[1] | |||
| C8H6N2OS2 | |||
| Molar mass | 210.27 g·mol−1 | ||
| Appearance | White to beige crystalline powder | ||
| Melting point | 133 °C (271 °F; 406 K) | ||
| Boiling point | 267 °C (513 °F; 540 K) | ||
| 7.7 mg/L (20 °C) | |||
| log P | 3.1 | ||
| Hazards[2] | |||
| GHS labelling: | |||
 
| |||
| Warning | |||
| H315, H317, H319, H335, H410 | |||
| P261, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P333+P313, P337+P313, P362, P363, P391, P403+P233, P405, P501 | |||
| Related compounds | |||
Related compounds
|
1,2,3-benzothiadiazole-7-carboxylic acid | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Acibenzolar-S-methyl is the ISO common name[3] for an organic compound that is used as a fungicide. Unusually, it is not directly toxic to fungi but works by inducing systemic acquired resistance, the natural defence system of plants.[4][5][6]
History
[edit]In the 1980s, researchers at Ciba-Geigy in Switzerland were seeking novel fungicides. They discovered that the methyl ester of 1,2,3-benzothiadiazole-7-carboxylic acid, and many other derivatives, had useful activity on fungal diseases, for example Pyricularia oryzae on rice.[7] In subsequent studies it was shown that the compound responsible for the biological activity was the carboxylic acid itself but that for optimum activity when used commercially it was important to choose a derivative which met requirements of product safety, ease of application and appropriate physical properties for translocation in the crop. After many derivatives of the acid had been tested, the S-methyl thioester was chosen for development under the code name CGA245704.[8] The product was launched in 1996 and is now sold by Syngenta with brand names including Bion[9] and Actigard.[10]
Synthesis
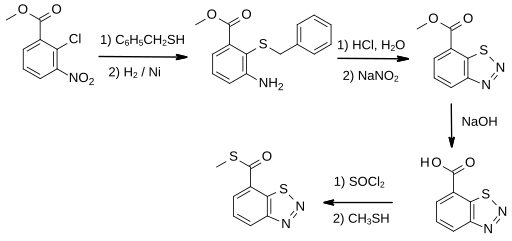
[edit]The first synthesis of the parent 1,2,3-benzothiadiazole-7-carboxylic acid was disclosed in patents filed by Ciba-Geigy.[7] The heterocyclic ring of the 1,2,3-benzothiadiazole core is formed by the classic ring-closure of a thiol onto a diazonium group adjacent in its benzene ring.[11] One example from the patent starts with methyl 2-chloro-3-nitrobenzoate, which reacts with benzyl mercaptan to give a thioether, which is converted by catalytic hydrogenation using Raney nickel to 3-amino-2-benzylthiobenzoic acid methyl ester. This intermediate, as its hydrochloride salt, is treated with sodium nitrite in water to give 7-methoxycarbonyl-1,2,3-benzothiadiazole in 86% yield after recrystallization.
Conversion of the ester into the thioester is by hydrolysis to form the free carboxylic acid followed by its conversion to the S-methyl thioester in a standard chemical transformation via the acid chloride.[7]
Mechanism of action
[edit]Acibenzolar-S-methyl has an unusual mechanism of action for a fungicide. It is not directly toxic to the fungus but instead activates the natural defences of the crop in a manner similar to the known role of salicylic acid and methyl jasmonate.[5][6][12] The genes for systemic acquired resistance are induced and pathogenesis-related proteins are produced.[13] The thioester is a propesticide for its active carboxylic acid metabolite. This hydrolysis reaction occurs in the plant, catalyzed by methyl salicylate esterase.[14]
Usage
[edit]In the USA
[edit]Pesticides are required to seek registration from appropriate authorities in the country in which they will be used.[15] In the United States, the Environmental Protection Agency (EPA) is responsible for regulating pesticides under the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA) and the Food Quality Protection Act (FQPA).[16] A pesticide can only be used legally according to the directions on the label that is included at the time of the sale of the pesticide. The purpose of the label is "to provide clear directions for effective product performance while minimizing risks to human health and the environment". A label is a legally binding document that mandates how the pesticide can and must be used and failure to follow the label as written when using the pesticide is a federal offence.[17]
Acibenzolar-S-methyl is registered in the US for use either as a seed treatment or for direct spraying on crops. Since it activates the crop's own defence mechanisms, it is not usually effective in curing established disease, only in protecting against future disease development. As a seed treatment it is registered for use on cotton, sunflower and sorghum while for spraying it is licensed on a wide range of vegetable crops.[18][19] The estimated annual use of acibenzolar-S-methyl in US agriculture is mapped by the US Geological Survey and shows a growing trend from its introduction in 2000 to 2017, the latest date for which figures are available. However, the total annual use has never exceeded 8000 lb (3600 kg), which is very low for an agrochemical. As expected given the main use on fruit and vegetables, the main areas of use are in California and Florida.[20]
In Europe
[edit]The product is also registered in Europe, where there is a monitoring programme to make sure that residues in food are below the limits set by the European Food Safety Authority. It is sometimes mixed with other pesticides to provide an extra degree of control by activating the crop's defence mechanisms in addition to the lethal effect of the main ingredient.
Human safety
[edit]Acibenzolar-S-methyl has little toxicity to mammals with an LD50 of over 2000 mg/kg (rats, oral).[1] However, it can cause moderate eye irritation. First aid information is included with the label.[19] The Codex Alimentarius database maintained by the FAO lists the maximum residue limits for acibenzolar-S-methyl and its parent acid in various food products.[21]
Resistance management
[edit]Although fungal populations have the ability to develop resistance to fungicides, the mechanism of action of acibenzolar-S-methyl gives it an advantage over conventional pesticides owing to its lack of direct toxicity to fungi. Nevertheless, regulatory bodies such as the EPA and the Fungicides Resistance Action Committee (FRAC)[22] monitor the risks of resistance developing: FRAC has assigned acibenzolar-S-methyl into its own class (group P01 fungicide).[23]
References
[edit]- ^ a b Pesticide Properties Database. "Acibenzolar-S-methyl". University of Hertfordshire.
- ^ "Acibenzolar-S-Methyl". US National Library of Medicine. Retrieved 2020-09-07.
- ^ "Compendium of Pesticide Common Names: acibenzolar". BCPC.
- ^ "Fact Sheet: Acibenzolar-S-Methyl" (PDF). EPA. 2000-08-11. Retrieved 2020-09-03.
- ^ a b Vlot, A.C.; Klessig, D.F.; Park, S.W. (August 2008). "Systemic acquired resistance, the elusive signal(s)". Current Opinion in Plant Biology. 11 (4): 436–442. Bibcode:2008COPB...11..436V. doi:10.1016/j.pbi.2008.05.003. hdl:11858/00-001M-0000-0012-36EC-0. PMID 18614393.
- ^ a b Gozzo, Franco; Faoro, Franco (2013). "Systemic Acquired Resistance (50 Years after Discovery): Moving from the Lab to the Field". Journal of Agricultural and Food Chemistry. 61 (51): 12473–12491. doi:10.1021/jf404156x. PMID 24328169.
- ^ a b c US patent 4931581, Schurter, R.; Kunz, W. & Nyfeler, R., "Process and a composition for immunizing plants against diseases", issued 1990-06-05, assigned to Ciba-Geigy Corporation
- ^ Kunz, W.; Schurter, R.; Maetzke, T. (September 1996). "The Chemistry of Benzothiadiazole Plant Activators". Pesticide Science. 50 (4): 275–282. doi:10.1002/(SICI)1096-9063(199708)50:4<275::AID-PS593>3.0.CO;2-7.
- ^ Syngenta US. "Bion 500 FS Seed Treatment". Retrieved 2020-09-04.
- ^ Syngenta US. "Actigard 40WG fungicide". Retrieved 2020-09-04.
- ^ Storr; Gilchrist, eds. (2004). "Product Class 9: 1,2,3-Thiadiazoles". Category 2, Hetarenes and Related Ring Systems. doi:10.1055/sos-SD-013-00386. ISBN 9783131122810.
- ^ Ryals JA, Neuenschwander UH, Willits MG, Molina A, Steiner HY, Hunt MD (October 1996). "Systemic Acquired Resistance". The Plant Cell. 8 (10): 1809–1819. doi:10.1105/tpc.8.10.1809. PMC 161316. PMID 12239363.
- ^ Cooper, Bret; Beard, Hunter S.; Garrett, Wesley M.; Campbell, Kimberly B. (2020). "Benzothiadiazole Conditions the Bean Proteome for Immunity to Bean Rust". Molecular Plant-Microbe Interactions. 33 (4): 600–611. doi:10.1094/MPMI-09-19-0250-R. PMID 31999214.
- ^ Jeschke, Peter (2016). "Propesticides and their use as agrochemicals". Pest Management Science. 72 (2): 210–225. doi:10.1002/ps.4170. PMID 26449612.
- ^ Willson HR (1996). "Pesticide Regulations". In Radcliffe EB, Hutchison WD, Cancelado RE (eds.). Radcliffe's IPM World Textbook. St. Paul: University of Minnesota. Archived from the original on July 13, 2017.
- ^ "Pesticides and Public Health". Pesticides: Health and Safety. US EPA. 2015-08-20. Archived from the original on January 14, 2014. Retrieved 2020-02-04.
- ^ EPA (2013-02-27). "The Pesticide Label". Retrieved 2020-02-02.
- ^ Syngenta US. "Bion 500FS (label)". Retrieved 2020-09-04.
- ^ a b Syngenta US. "Actigard plant activator (label)". Retrieved 2020-09-04.
- ^ US Geological Survey (2020-06-18). "Estimated Agricultural Use for Acibenzolar, 2017". Retrieved 2020-09-05.
- ^ FAO / WHO (2017). "Acibenzolar-S-methyl". Retrieved 2020-09-04.
- ^ "Fungicides Resistance Action Committee website".
- ^ "Fungal control agents sorted by cross resistance pattern and mode of action" (PDF). 2020. Archived from the original (PDF) on 2021-08-16. Retrieved 2020-09-04.
Further reading
[edit]- Morton, V.; Staub, T. (2008). "A Short History of Fungicides". Apsnet Feature Articles. doi:10.1094/APSnetFeature-2008-0308.
External links
[edit]- Acibenzolar-S-methyl in the Pesticide Properties DataBase (PPDB)