Template:Infobox drug/doc
| This is a documentation subpage for Template:Infobox drug. It may contain usage information, categories and other content that is not part of the original template page. |
| This template is used on approximately 9,800 pages and changes may be widely noticed. Test changes in the template's /sandbox or /testcases subpages, or in your own user subpage. Consider discussing changes on the talk page before implementing them. |
| This template adds an automatically generated short description. If the automatic short description is not optimal, replace it by adding {{Short description}} at the top of the article. |
| This template uses Lua: |
{{Infobox drug}} or {{Drugbox}} is the infobox for drugs, both medical and recreational. It can be used for single chemicals (most common), and for #Combination products, #Monoclonal antibody drugs and #Vaccines.
Type of drug
[edit]Single chemical (type=)
[edit]Drugs that are a simple chemical compound (about 90% of the drug articles are). Short parameter list:
{{Infobox drug
| drug_name =
| INN =
| type = <!-- empty -->
| image =
| alt =
| caption =
<!-- Clinical data -->
| pronounce =
| tradename =
| Drugs.com =
| MedlinePlus =
| pregnancy_AU = <!-- A / B1 / B2 / B3 / C / D / X -->
| pregnancy_AU_comment =
| pregnancy_category=
| routes_of_administration =
| ATCvet =
| ATC_prefix = <!-- 'none' if uncategorised -->
| ATC_suffix =
<!-- Legal status -->
| legal_AU = <!-- S2, S3, S4, S5, S6, S7, S8, S9 or Unscheduled -->
| legal_AU_comment =
| legal_BR = <!-- OTC, A1, A2, A3, B1, B2, C1, C2, C3, C5, D1, D2, E, F1, F2, F3, F4 -->
| legal_BR_comment =
| legal_CA = <!-- OTC, Rx-only, Schedule I, II, III, IV, V, VI, VII, VIII -->
| legal_CA_comment =
| legal_DE = <!-- Anlage I, II, III or Unscheduled -->
| legal_DE_comment =
| legal_NZ = <!-- Class A, B, C -->
| legal_NZ_comment =
| legal_UK = <!-- GSL, P, POM, CD, CD Lic, CD POM, CD No Reg POM, CD (Benz) POM, CD (Anab) POM or CD Inv POM / Class A, B, C -->
| legal_UK_comment =
| legal_US = <!-- OTC / Rx-only / Schedule I, II, III, IV, V -->
| legal_US_comment =
| legal_EU =
| legal_EU_comment =
| legal_UN = <!-- N I, II, III, IV / P I, II, III, IV -->
| legal_UN_comment =
| legal_status = <!-- For countries not listed above -->
<!-- Pharmacokinetic data -->
| bioavailability =
| protein_bound =
| metabolism =
| metabolites =
| onset =
| elimination_half-life =
| duration_of_action=
| excretion =
<!-- Identifiers -->
| CAS_number =
| PubChem =
| DrugBank =
<!-- Chemical and physical data -->
| IUPAC_name =
| chemical_formula =
| molecular_weight =
}}
Full single-drug template with extended fields:
{{Infobox drug
| drug_name =
| INN =
| type = <!-- empty -->
| image =
| width =
| alt =
| caption =
| image2 =
| width2 =
| alt2 =
| caption2 =
| imageL =
| widthL =
| altL =
| imageR =
| widthR =
| altR =
| captionLR =
<!-- Clinical data -->
| pronounce =
| tradename =
| Drugs.com =
| MedlinePlus =
| licence_CA = <!-- Health Canada may use generic or brand name (generic name preferred) -->
| licence_EU = <!-- EMA uses INN (or special INN_EMA) -->
| DailyMedID = <!-- DailyMed may use generic or brand name (generic name preferred) -->
| licence_US = <!-- FDA may use generic or brand name (generic name preferred) -->
| pregnancy_AU = <!-- A / B1 / B2 / B3 / C / D / X -->
| pregnancy_AU_comment =
| pregnancy_category=
| tolerance_potential =
| dependency_liability =
| addiction_liability =
| routes_of_administration =
| class =
| ATCvet =
| ATC_prefix = <!-- 'none' if uncategorised -->
| ATC_suffix =
| ATC_supplemental =
<!-- Legal status -->
| legal_AU = <!-- S2, S3, S4, S5, S6, S7, S8, S9 or Unscheduled -->
| legal_AU_comment =
| legal_BR = <!-- OTC, A1, A2, A3, B1, B2, C1, C2, C3, C5, D1, D2, E, F1, F2, F3, F4 -->
| legal_BR_comment =
| legal_CA = <!-- OTC, Rx-only, Schedule I, II, III, IV, V, VI, VII, VIII -->
| legal_CA_comment =
| legal_DE = <!-- Anlage I, II, III or Unscheduled -->
| legal_DE_comment =
| legal_NZ = <!-- Class A, B, C -->
| legal_NZ_comment =
| legal_UK = <!-- GSL, P, POM, CD, CD Lic, CD POM, CD No Reg POM, CD (Benz) POM, CD (Anab) POM or CD Inv POM / Class A, B, C -->
| legal_UK_comment =
| legal_US = <!-- OTC / Rx-only / Schedule I, II, III, IV, V -->
| legal_US_comment =
| legal_EU =
| legal_EU_comment =
| legal_UN = <!-- N I, II, III, IV / P I, II, III, IV -->
| legal_UN_comment =
| legal_status = <!-- For countries not listed above -->
<!-- Pharmacokinetic data -->
| bioavailability =
| protein_bound =
| metabolism =
| metabolites =
| onset =
| elimination_half-life =
| duration_of_action=
| excretion =
<!-- Identifiers -->
| CAS_number =
| CAS_supplemental =
| PubChem =
| PubChemSubstance =
| IUPHAR_ligand =
| DrugBank =
| ChemSpiderID =
| UNII =
| KEGG =
| ChEBI =
| ChEMBL =
| NIAID_ChemDB =
| PDB_ligand =
| synonyms =
<!-- Chemical and physical data -->
| IUPAC_name =
| chemical_formula =
| C= | H= | Ag= | Al= | As= | Au= | B= | Bi= | Br= | Ca= | Cl= | Co= | F= | Fe= | Gd= | I=
| K= | Li= | Mg= | Mn= | N= | Na= | O= | P= | Pt= | S= | Sb= | Se= | Sr= | Tc= | Zn= | charge=
| molecular_weight =
| SMILES =
| Jmol =
| StdInChI =
| StdInChI_comment =
| StdInChIKey =
| density =
| density_notes =
| melting_point =
| melting_high =
| melting_notes =
| boiling_point =
| boiling_notes =
| solubility =
| sol_units =
| specific_rotation =
}}
Automatic generation
[edit]The Wikipedia Drugbox and Chembox Maker at the Wayback Machine (archived June 2, 2023) can be used to automatically generate single-chemical drugbox templates. Note: For unknown reason the For quicker access, this tool can be added to the tools section of Wikipedia's left sidebar via User:Qwerfjkl/scripts/chemboxmaker.
Another option is Diberri and Boghog's excellent Wikipedia template filling tool which will generate a populated template to copy and paste into an article if "DrugBank ID" is selected in the dropdown menu and a DrugBank ID number (which may be obtained from DrugBank search) is added in the adjacent field. Please select for Fill template vertically, but as Wikipedia is a general encyclopedia, most drugs do not warrant Show extended fields. However, drugbox generation with this tool is currently broken and has been for some time.
Combination of drugs (type=combo)
[edit]
For drug-combinations, at least two components must be defined, with more optional (up to 6). For each component:
| component''n'' = <!-- name, will be wikilinked -->
| class''n'' = <!-- class of agent, needs manual wikilinking -->
Example for Seretide:
| Combination of | |
|---|---|
| Fluticasone | Glucocorticoid |
| Salmeterol | Long-acting beta2 agonist |
| drug_name = Seretide
| type = combo
<!-- Combo data -->
| component1 = Fluticasone
| class1 = [[Glucocorticoid]]
| component2 = Salmeterol
| class2 = [[Beta2-adrenergic receptor agonist|Long-acting beta2 agonist]]
Generally combination articles will not need to display the molecular images of its constituents (the relevant specific articles would have the images). It disables all Chemical & Pharmacology parameters (which describe properties of single drug items). These redundant disabled parameters are best not included in the template calling, so use the following abridged forms of the template:
Shortened combination product form:
{{Infobox drug
| drug_name =
| type = combo
<!-- Combo data -->
| component1 = <!-- Drugname, automatically linked -->
| class1 = <!-- Group, manual link using [[..|..]] -->
| component2 = <!-- Drugname, automatically linked -->
| class2 = <!-- Group, manual link using [[..|..]] -->
<!-- Clinical data -->
| pronounce =
| tradename =
| Drugs.com =
| MedlinePlus =
| pregnancy_AU = <!-- A / B1 / B2 / B3 / C / D / X -->
| pregnancy_AU_comment =
| pregnancy_category=
| routes_of_administration =
| ATCvet =
| ATC_prefix = <!-- 'none' if uncategorised -->
| ATC_suffix =
<!-- Legal status -->
| legal_AU = <!-- S2, S3, S4, S5, S6, S7, S8, S9 or Unscheduled -->
| legal_AU_comment =
| legal_BR = <!-- OTC, A1, A2, A3, B1, B2, C1, C2, C3, C5, D1, D2, E, F1, F2, F3, F4 -->
| legal_BR_comment =
| legal_CA = <!-- OTC, Rx-only, Schedule I, II, III, IV, V, VI, VII, VIII -->
| legal_CA_comment =
| legal_DE = <!-- Anlage I, II, III or Unscheduled -->
| legal_DE_comment =
| legal_NZ = <!-- Class A, B, C -->
| legal_NZ_comment =
| legal_UK = <!-- GSL, P, POM, CD, CD Lic, CD POM, CD No Reg POM, CD (Benz) POM, CD (Anab) POM or CD Inv POM / Class A, B, C -->
| legal_UK_comment =
| legal_US = <!-- OTC / Rx-only / Schedule I, II, III, IV, V -->
| legal_US_comment =
| legal_EU =
| legal_EU_comment =
| legal_UN = <!-- N I, II, III, IV / P I, II, III, IV -->
| legal_UN_comment =
| legal_status = <!-- For countries not listed above -->
<!-- Identifiers -->
| CAS_number =
| PubChem =
| DrugBank =
| KEGG =
}}
Full combination product template with extended fields and allowing for up to six items:
{{Infobox drug
| drug_name =
| type = combo
<!-- Combo data -->
| component1 = <!-- Drugname, automatically linked -->
| class1 = <!-- Group, manual link using [[..|..]] -->
| component2 = <!-- Drugname, automatically linked -->
| class2 = <!-- Group, manual link using [[..|..]] -->
| component3 = <!-- Drugname, automatically linked -->
| class3 = <!-- Group, manual link using [[..|..]] -->
| component4 = <!-- Drugname, automatically linked -->
| class4 = <!-- Group, manual link using [[..|..]] -->
| component5 = <!-- Drugname, automatically linked -->
| class5 = <!-- Group, manual link using [[..|..]] -->
| component6 = <!-- Drugname, automatically linked -->
| class6 = <!-- Group, manual link using [[..|..]] -->
<!-- Clinical data -->
| pronounce =
| tradename =
| Drugs.com =
| MedlinePlus =
| licence_CA = <!-- Health Canada may use generic or brand name (generic name preferred) -->
| licence_EU = <!-- EMA uses INN (or special INN_EMA) -->
| DailyMedID = <!-- DailyMed may use generic or brand name (generic name preferred) -->
| licence_US = <!-- FDA may use generic or brand name (generic name preferred) -->
| pregnancy_AU = <!-- A / B1 / B2 / B3 / C / D / X -->
| pregnancy_AU_comment =
| pregnancy_category=
| dependency_liability =
| addiction_liability =
| routes_of_administration =
| ATCvet =
| ATC_prefix = <!-- 'none' if uncategorised -->
| ATC_suffix =
| ATC_supplemental =
<!-- Legal status -->
| legal_AU = <!-- S2, S3, S4, S5, S6, S7, S8, S9 or Unscheduled -->
| legal_AU_comment =
| legal_BR = <!-- OTC, A1, A2, A3, B1, B2, C1, C2, C3, C5, D1, D2, E, F1, F2, F3, F4 -->
| legal_BR_comment =
| legal_CA = <!-- OTC, Rx-only, Schedule I, II, III, IV, V, VI, VII, VIII -->
| legal_CA_comment =
| legal_DE = <!-- Anlage I, II, III or Unscheduled -->
| legal_DE_comment =
| legal_NZ = <!-- Class A, B, C -->
| legal_NZ_comment =
| legal_UK = <!-- GSL, P, POM, CD, CD Lic, CD POM, CD No Reg POM, CD (Benz) POM, CD (Anab) POM or CD Inv POM / Class A, B, C -->
| legal_UK_comment =
| legal_US = <!-- OTC / Rx-only / Schedule I, II, III, IV, V -->
| legal_US_comment =
| legal_EU =
| legal_EU_comment =
| legal_UN = <!-- N I, II, III, IV / P I, II, III, IV -->
| legal_UN_comment =
| legal_status = <!-- For countries not listed above -->
<!-- Identifiers -->
| CAS_number =
| CAS_supplemental =
| PubChem =
| PubChemSubstance =
| IUPHAR_ligand =
| DrugBank =
| ChemSpiderID =
| UNII =
| KEGG =
| ChEBI =
| ChEMBL =
| synonyms =
}}
Monoclonal antibody drugs (type=mab)
[edit]
|type=mab
This form the box uses parameters:
| type = mab
<!-- Monoclonal antibody data -->
| mab_type =
| source =
| target =
For example Catumaxomab:
| Monoclonal antibody | |
|---|---|
| Type | Trifunctional antibody |
| Source | Rat/mouse hybrid |
| Target | EpCAM, CD3 |
| type = mab
<!-- Monoclonal antibody data -->
| mab_type = 3funct
| source = axo
| target = [[EpCAM]], [[CD3 (immunology)|CD3]]
mab_type defines one of the following types of antibody or antibody fragment:
|mab_type= |
output |
|---|---|
| mab | Whole antibody |
| Fab | Fab fragment |
| F(ab')2 | F(ab')2 fragment |
| Fab' | Fab' fragment |
| scFv | Single-chain variable fragment |
| di-scFv | Di-single-chain variable fragment |
| sdAb | Single domain antibody |
| 3funct | Trifunctional antibody |
| clFab | Chemically linked Fab |
| BiTE | Bi-specific T-cell engager |
source is the species on which the protein sequence of the antibody is based:
|source= |
output |
|---|---|
| a | Rat |
| e | Hamster |
| i | Primate |
| o | Mouse |
| u | Human |
| xi/a, xi/e, etc. | Chimeric (rat/human) etc. |
| xi | Chimeric [generic, use only if values above are not applicable] |
| zu/a, zu/e, etc. | Humanized (from rat) etc. |
| zu | Humanized [generic, use only if values above are not applicable] |
| xizu/a, xizu/e, etc. | Chimeric/humanized hybrid (rat/human) etc. |
| xizu | Chimeric/humanized hybrid [generic, use only if values above are not applicable] |
| axo | Rat/mouse hybrid |
target is the antigen at which the antibody is directed. This parameter takes free text as value, preferably including a wikilink such as |target=[[TNF-α]].
The drug name is followed by a "?" linked to Nomenclature of monoclonal antibodies, saving the need to explain how each monoclonal antibody has been named.
Shortened Monoclonal antibody form:
{{Infobox drug
| type = mab
| image =
| alt =
| caption =
<!-- Monoclonal antibody data -->
| mab_type = <!-- mab, Fab, F(ab')2, Fab', scFv, di-scFv, 3funct, clFab, BiTE -->
| source = <!-- a, e, i, o, u, xi/a, zu/a, xizu/a, axo, ... -->
| target = <!-- antigen, not the code number of the monoclonal antibody! -->
<!-- Clinical data -->
| pronounce =
| tradename =
| Drugs.com =
| MedlinePlus =
| pregnancy_AU = <!-- A / B1 / B2 / B3 / C / D / X -->
| pregnancy_AU_comment =
| pregnancy_category=
| routes_of_administration =
| ATC_prefix = <!-- 'none' when not assigned -->
| ATC_suffix =
<!-- Legal status -->
| legal_AU = <!-- S2, S3, S4, S5, S6, S7, S8, S9 or Unscheduled -->
| legal_AU_comment =
| legal_BR = <!-- OTC, A1, A2, A3, B1, B2, C1, C2, C3, C5, D1, D2, E, F1, F2, F3, F4 -->
| legal_BR_comment =
| legal_CA = <!-- OTC, Rx-only, Schedule I, II, III, IV, V, VI, VII, VIII -->
| legal_CA_comment =
| legal_DE = <!-- Anlage I, II, III or Unscheduled -->
| legal_DE_comment =
| legal_NZ = <!-- Class A, B, C -->
| legal_NZ_comment =
| legal_UK = <!-- GSL, P, POM, CD, CD Lic, CD POM, CD No Reg POM, CD (Benz) POM, CD (Anab) POM or CD Inv POM / Class A, B, C -->
| legal_UK_comment =
| legal_US = <!-- OTC / Rx-only / Schedule I, II, III, IV, V -->
| legal_US_comment =
| legal_EU =
| legal_EU_comment =
| legal_UN = <!-- N I, II, III, IV / P I, II, III, IV -->
| legal_UN_comment =
| legal_status = <!-- For countries not listed above -->
<!-- Pharmacokinetic data -->
| bioavailability =
| protein_bound =
| metabolism =
| metabolites =
| onset =
| elimination_half-life =
| duration_of_action=
| excretion =
<!-- Identifiers -->
| CAS_number =
| PubChem =
| DrugBank =
| KEGG =
}}
Vaccines (type=vaccine)
[edit]|type=vaccine
This allows the alternative parameters of the vaccine_type and target to be specified:
- target can be any text. Can be used to link to a specific article
- vaccine_type from the defined list of options as described at Vaccine#Types, the infobox will provide standardised display and links as follows:
| vaccine_type value | Output |
|---|---|
| inactivated or killed | Inactivated |
| attenuated | Attenuated |
| subunit | Subunit |
| protein or protein subunit | Protein subunit |
| peptide or peptide subunit | Peptide subunit |
| polysaccharide | Polysaccharide |
| vlp | Virus-like particles |
| toxoid | Toxoid |
| conjugate | Conjugate |
| viral | Viral vector |
| recombinant | Recombinant vector |
| dna | DNA |
| rna | RNA |
| mrna | mRNA |
| heterologous | Heterologous |
| any text | any text (shows unedited) |
Where a vaccination product protects against more than one agent, please use the combination form of this infobox with type=combo (per #Combination products), and each classX=[[Vaccine]].
Shortened Vaccine form:
{{Infobox drug
| drug_name =
| type = vaccine
| image =
| alt =
| caption =
<!-- Vaccine data -->
| target = <!-- antigen/bacteria/toxin/virus -->
| vaccine_type = <!-- killed/attenuated/live/toxoid/protein subunit/subunit/conjugate/recombinant/DNA -->
<!-- Clinical data -->
| pronounce =
| tradename =
| Drugs.com =
| MedlinePlus =
| pregnancy_AU = <!-- A / B1 / B2 / B3 / C / D / X -->
| pregnancy_AU_comment =
| pregnancy_category=
| routes_of_administration =
| ATCvet =
| ATC_prefix = <!-- 'none' if uncategorised -->
| ATC_suffix =
<!-- Legal status -->
| legal_AU = <!-- S2, S3, S4, S5, S6, S7, S8, S9 or Unscheduled -->
| legal_AU_comment =
| legal_BR = <!-- OTC, A1, A2, A3, B1, B2, C1, C2, C3, C5, D1, D2, E, F1, F2, F3, F4 -->
| legal_BR_comment =
| legal_CA = <!-- OTC, Rx-only, Schedule I, II, III, IV, V, VI, VII, VIII -->
| legal_CA_comment =
| legal_DE = <!-- Anlage I, II, III or Unscheduled -->
| legal_DE_comment =
| legal_NZ = <!-- Class A, B, C -->
| legal_NZ_comment =
| legal_UK = <!-- GSL, P, POM, CD, CD Lic, CD POM, CD No Reg POM, CD (Benz) POM, CD (Anab) POM or CD Inv POM / Class A, B, C -->
| legal_UK_comment =
| legal_US = <!-- OTC / Rx-only / Schedule I, II, III, IV, V -->
| legal_US_comment =
| legal_EU =
| legal_EU_comment =
| legal_UN = <!-- N I, II, III, IV / P I, II, III, IV -->
| legal_UN_comment =
| legal_status = <!-- For countries not listed above -->
<!-- Identifiers -->
| CAS_number =
| PubChem =
| DrugBank =
}}
Full Vaccine template with extended fields:
{{Infobox drug
| drug_name =
| type = vaccine
| image =
| alt =
| width =
| caption =
| image2 =
| alt2 =
| width2 =
| caption2 =
<!-- Vaccine data -->
| target = <!-- the antigen/bacteria/toxin/virus to protect against -->
| vaccine_type = <!-- killed/attenuated/live/toxoid/protein subunit/subunit/conjugate/recombinant/DNA -->
<!-- Clinical data -->
| pronounce =
| tradename =
| Drugs.com =
| MedlinePlus =
| licence_CA = <!-- Health Canada may use generic or brand name (generic name preferred) -->
| licence_EU = <!-- EMA uses INN (or special INN_EMA) -->
| DailyMedID = <!-- DailyMed may use generic or brand name (generic name preferred) -->
| licence_US = <!-- FDA may use generic or brand name (generic name preferred) -->
| pregnancy_AU = <!-- A / B1 / B2 / B3 / C / D / X -->
| pregnancy_AU_comment =
| pregnancy_category=
| routes_of_administration =
| ATCvet =
| ATC_prefix = <!-- 'none' if uncategorised -->
| ATC_suffix =
<!-- Legal status -->
| legal_AU = <!-- S2, S3, S4, S5, S6, S7, S8, S9 or Unscheduled -->
| legal_AU_comment =
| legal_BR = <!-- OTC, A1, A2, A3, B1, B2, C1, C2, C3, C5, D1, D2, E, F1, F2, F3, F4 -->
| legal_BR_comment =
| legal_CA = <!-- OTC, Rx-only, Schedule I, II, III, IV, V, VI, VII, VIII -->
| legal_CA_comment =
| legal_DE = <!-- Anlage I, II, III or Unscheduled -->
| legal_DE_comment =
| legal_NZ = <!-- Class A, B, C -->
| legal_NZ_comment =
| legal_UK = <!-- GSL, P, POM, CD, CD Lic, CD POM, CD No Reg POM, CD (Benz) POM, CD (Anab) POM or CD Inv POM / Class A, B, C -->
| legal_UK_comment =
| legal_US = <!-- OTC / Rx-only / Schedule I, II, III, IV, V -->
| legal_US_comment =
| legal_EU =
| legal_EU_comment =
| legal_UN = <!-- N I, II, III, IV / P I, II, III, IV -->
| legal_UN_comment =
| legal_status = <!-- For countries not listed above -->
<!-- Identifiers -->
| CAS_number =
| CAS_supplemental =
| PubChem =
| PubChemSubstance =
| IUPHAR_ligand =
| DrugBank =
| ChemSpiderID =
| UNII =
| KEGG =
| ChEBI =
| ChEMBL =
| NIAID_ChemDB =
}}
Parameters
[edit]Full parameter list
[edit]{{Infobox drug}} parameter list
|
|---|
|
This parameter list:
<!-- TITLE ----- ----- -->
| drug_name =
| INN =
| type = <!-- drugtype, can be: =combo, =mab, =vaccine, =<blank> (for simple chemical). -->
<!-- IMAGES ----- ----- -->
| image =
| width =
| alt =
| caption =
| image2 =
| width2 =
| alt2 =
| caption2 =
| imageL =
| imageR =
| widthL =
| widthR =
| altL =
| altR =
| captionLR =
<!-- type=VACCINE: ----- ----- -->
| target =
| vaccine_type =
<!-- type=MAB: ----- ----- -->
| mab_type =
| source =
| target =
<!-- type=COMBO: ----- ----- -->
| component1 =
| class =
| component2 =
| class =
| component3 =
| class3 =
| component4 =
| class4 =
| component5 =
| class5 =
<!-- GENE THERAPY ----- ----- -->
| gt_vector =
| gt_nucleic_acid_type =
| gt_editing_method =
| gt_delivery_method =
<!-- CLINICAL data ----- ----- -->
| pronounce =
| pronounce_comment =
| pronounce_ref =
| tradenames =
| synonyms =
| biosimilars =
| Drugs.com =
| MedlinePlus =
| licence_CA =
| licence_EU =
| DailyMedID =
| license_US = <!-- Both licenCe/licenSe spelling accepted; see also engvar= for spelling shown -->
| license_CA =
| license_EU =
| license_US =
| pregnancy_AU =
| pregnancy_US =
| pregnancy_AU_comment =
| pregnancy_US_comment =
| pregnancy_category =
| PLLR =
| legal_AU =
| legal_CA =
| legal_DE =
| legal_NZ =
| legal_UK =
| legal_US =
| legal_EU =
| legal_UN =
| legal_AU_comment =
| legal_CA_comment =
| legal_DE_comment =
| legal_NZ_comment =
| legal_UK_comment =
| legal_US_comment =
| legal_EU_comment =
| legal_UN_comment =
| legal_status =
| dependency_liability =
| addiction_liability =
| routes_of_administration =
<!-- PHYSIOLOGICAL ----- ----- -->
| source_tissues =
| target_tissues =
| receptors =
| agonists =
| antagonists =
| precursor =
| biosynthesis =
| metabolism = <!-- same parameter as in pharmacokinetic data -->
| AAN = <!-- INN variants -->
| BAN =
| JAN =
| USAN =
<!-- PHARMACOKINETIC ----- ----- -->
| bioavailability =
| protein_bound =
| metabolism =
| metabolites =
| onset =
| elimination_half-life =
| duration_of_action =
| excretion =
<!-- IDENTIFIERS ----- ----- -->
| IUPAC_name = <!-- when drugtype is single chemical -->
| index_label =
| index2_label =
| index_comment =
| index2_comment =
| CAS_number =
| CAS_supplemental =
| CAS_number2 =
| CAS_supplemental2 =
| class =
| ATCvet = <!-- can be: yes -->
| ATC_prefix =
| ATC_suffix =
| ATC_supplemental =
| ATC_prefix2 =
| ATC_suffix2 =
| PubChem =
| PubChem2 =
| PubChemSubstance =
| PubChemSubstance2 =
| IUPHAR_ligand =
| IUPHAR_ligand2 =
| DrugBank =
| DrugBank2 =
| ChemSpiderID =
| ChemSpiderID2 =
| UNII =
| UNII2 =
| KEGG =
| KEGG2 =
| ChEBI =
| ChEBI2 =
| ChEMBL =
| ChEMBL2 =
| NIAID_ChemDB =
| NIAID_ChemDB2 =
| PDB_ligand =
| PDB_ligand2 =
<!-- CHEMICAL data ----- ----- -->
| chemical_formula =
| Ac =
| Ag =
| Al =
| Am =
| Ar =
| As =
| At =
| Au =
| B =
| Ba =
| Be =
| Bh =
| Bi =
| Bk =
| Br =
| C =
| Ca =
| Cd =
| Ce =
| Cf =
| Cl =
| Cm =
| Cn =
| Co =
| Cr =
| Cs =
| Cu =
| Db =
| Ds =
| Dy =
| Er =
| Es =
| Eu =
| F =
| Fe =
| Fl =
| Fm =
| Fr =
| Ga =
| Gd =
| Ge =
| H =
| He =
| Hf =
| Hg =
| Ho =
| Hs =
| I =
| In =
| Ir =
| K =
| Kr =
| La =
| Li =
| Lr =
| Lu =
| Lv =
| Md =
| Mg =
| Mn =
| Mo =
| Mt =
| N =
| Na =
| Nb =
| Nd =
| Ne =
| Ni =
| No =
| Np =
| O =
| Os =
| P =
| Pa =
| Pb =
| Pd =
| Pm =
| Po =
| Pr =
| Pt =
| Pu =
| Ra =
| Rb =
| Ref =
| Rf =
| Rg =
| Rh =
| Rn =
| Ru =
| S =
| Sb =
| Sc =
| Se =
| Sg =
| Si =
| Sm =
| Sn =
| Sr =
| Ta =
| Tb =
| Tc =
| Te =
| Th =
| Ti =
| Tl =
| Tm =
| U =
| Nh =
| Mc =
| Ts =
| Og =
| V =
| W =
| Xe =
| Y =
| Yb =
| Zn =
| Zr =
| charge =
| chemical_formula_ref =
| chemical_formula_comment =
| molecular_weight =
| molecular_weight_round =
| molecular_weight_unit =
| molecular_weight_ref =
| molecular_weight_comment =
| chirality = <!-- could be: [[Racemic mixture]] -->
| specific_rotation =
| SMILES =
| smiles =
| SMILES2 =
| smiles2 =
| StdInChI =
| StdInChI_comment =
| StdInChIKey =
| StdInChI2 =
| StdInChIKey2 =
<!-- PHYSICAL data ----- ----- -->
| density =
| density_notes =
| melting_point =
| melting_high =
| boiling_point =
| boiling_high =
| solubility =
<!-- DATA PAGE ----- ----- -->
| data page = <!-- overrules default existing {PAGENAME} (data page) -->
<!-- VERIFICATIONS (bot-maintained) ----- ----- -->
| CAS_number_Ref =
| PubChem_Ref =
| DrugBank_Ref =
| ChemSpiderID_Ref =
| UNII_Ref =
| KEGG_Ref =
| ChEBI_Ref =
| ChEMBL_Ref =
| StdInChI_Ref =
| StdInChIKey_Ref =
| CAS_number2_Ref =
| PubChem2_Ref =
| DrugBank2_Ref =
| ChemSpiderID2_Ref =
| UNII2_Ref =
| KEGG2_Ref =
| ChEBI2_Ref =
| ChEMBL2_Ref =
| StdInChI2_Ref =
| StdInChIKey2_Ref =
| Verifiedfields =
| verifiedrevid =
Recent parameter changes[edit]This parameter list:
| gt_target_gene = RPI35
| gt_vector = adenovirus serotype 2
| gt_nucleic_acid_type = viral vector
| gt_editing_method = [[RPE65]]
| gt_delivery_method = [not applicable here]
<!-- Physiological data -->
| source_tissues =
| target_tissues =
| receptors =
| agonists =
| antagonists =
| precursor =
| biosynthesis =
| metabolism =
A second chemical has index #2.
|
Infobox title and INN
[edit]By default, the title of the infobox is the article title. You can overwrite that using
|drug_name=.
Per the Manual of Style, the infobox title must be the International nonproprietary name (INN). There is a tooltip text referring to INN.
When, for some reason, the title is not the INN, the correct INN can be added as a subtitle using |INN=
| drug_name = Heroin
| INN = Diamorphine
When the drug has no INN at all, setting |INN=none will suppress the tooltip text mentioning INN.
- See also #Names and identifiers
Images
[edit]You can use the image to provide an illustration. A caption can be added. The alt parameter is a textual description of the image, and is shown in the 'tooltip' and read by visually impaired readers (more in WP:ALT). Parameter width sets the width in px; default width is 200px.
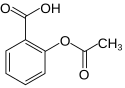 structure |
| image = Acetylsalicylsäure2.svg
| caption = structure
| alt = Molecule of aspirin
| width = 125
A second image can be added using image2:
| image2 =
| caption2 =
| alt2 =
| width2 =
Also a side-by-side pair of images can be added (with single caption):
| imageL =
| widthL =
| altL =
| imageR=
| widthR =
| altR =
| captionLR =
See also |SMILES= input for the 3D model by Jmol (an external link).
Clinical data
[edit]| tradename = | Drugs.com = | MedlinePlus = | routes_of_administration =
- tradename – comma separated list of trade names by the originator (no generics, not more than three names)
- Drugs.com – link to Drugs.com monograph
- MedlinePlus – MedlinePlus drug accession number
Pregnancy category
[edit]- See also pregnancy category, documentation for extensive documentation
Pregnancy Categorisation has been previously declared using just the pregnancy_category parameter with a variety of markup styles used to indicate various countries and their classifications. Alternatively pregnancy_AU may be set to the fixed categories. For Australia values permitted are: A, B1, B2, B3, C, D or X (note if just set to 'B' then B? is shown). Pregnancy categories are no longer used in the US.
Note the UK does not have official designated categorisations, but has both a variety of standard phrases with explanatory qualifications, plus many individual descriptions. The generic |pregnancy_category= accepts any text, and may be used with such country-specific information (non AU or US).
| Clinical data | |
|---|---|
| Pregnancy category |
|
| pregnancy_AU = B1
| pregnancy_AU_comment = <ref>...</ref>
| pregnancy_category = Used in pregnancy against [[Pregnancy-induced hypertension|PIH]]
Dependence liability
[edit]Optional parameters Dependence liability and Addiction_liability allow opioids or benzodiazepines to be flagged with the risk of becoming dependent/addicted upon them, although in many cases this may be somewhat subjective. Drugs should be rated as Low, Medium, High or Extremely high:
| dependency_liability = High
| addiction_liability = High
Licensing information
[edit]There are three major licensing authorities that make drug information online accessible: the European Medicines Agency (EMA, EU), and the Food and Drug Administration (FDA, US).
The FDA "Label Information" and the EMA "Product Information", where available, are very detailed. There is material aimed at the professional and also plain-English consumer information. These are excellent reliable source for article information but also contains much that makes it a worthwhile "External link" since there is no way one can include all the info. Both sites work when using the brand name of the drug but the FDA site also allows searches with the generic name (active ingredient), which lists generic variants where they are licensed. Health Canada's site operates similarly to the FDA's site, with Canada-specific drug monographs and other information. The DailyMed site is a more user-friendly repository of primarily labeling information. For all sites, the use of the generic name of the drug is preferred whenever possible as this will list all brand and generic forms of the drug.
| licence_EU = <!-- Any input here (like '=yes'), will give link by INN -->
| DailyMedID = <!-- DailyMed may use generic or brand name (generic name preferred) -->
| licence_US = <!-- or |license_US=; FDA may use generic or brand name (generic name preferred) -->
|licence_EU=yeswill create a link to the EMA site, using the INN (i.e., INN, drug_name or article title). When the EMA used INN is different, enter its INN in|INN_EMA=.
Lists of products and the parameter term to use may be easily searched for:
- European Public Assessment Reports: SiteMap, authorised products A–Z: human, veterinary
- Licence EU (EMA): any input in
|licence_EU=will give a link to the EMA page for the active substance (INN). - Drugs@FDA Search by Drug Name or Active Ingredient
- Where items are composed of more than one word, DailyMedID and licence_US require an underscore '_' in place of any spaces; e.g. Acyclovir_sodium.
Drug class
[edit]| class = any text
The drug class designates the family that this drug belongs to. Typically the chemical class (e.g., benzodiazepine), mechanism of action (e.g., beta blocker), mode of action (e.g., diuretic), and/or therapeutic class (e.g., analgesic). Hence this field may contain more than one drug class if appropriate (each preferably wiki linked).
ATC, ATCvet
[edit]Where the drug is not included in ATC coding system (as opposed to its value just not been entered into the template) then set ATC_prefix to 'none', and the article is automatically added to Category:Drugs not assigned an ATC code:
| ATC_prefix = none
Additional ATC(vet) may be included with the optional |ATC_supplemental= parameter as a comma-separated list. ATC templates may be used to have these additional codes link to the relevant databases:
| ATC_supplemental ={{ATC|M02|AA15}}, {{ATCvet|S01|BC03}}
Adds the following linked codes:
Veterinary drugs are placed in a slightly different classification system, ATCvet. The code may be specified as ATCvet by setting the parameter ATCvet to 'yes'. Do not include the leading 'Q' in ATC_prefix:
| ATCvet = yes
| ATC_prefix = N05
| ATC_suffix = AX90
When the ATCvet code happens to be the same as the ATC for a mainly human drug, it is usually a better idea to just provide the ATC in the infobox.
Legal status
[edit]- See also: {{Infobox drug/legal status}} for extensive documentation.
Legal status allows to specify which controlling acts are active in various countries and organisations. Available parameters are |legal_AU, legal_BR, legal_CA, legal_DE, legal_NZ, legal_UK, legal_US, legal_EU, legal_UN=, and links are provided for standard input. Also, the parameters can have the suffix |..._comment=.
Also available is |legal_status=, which is general and allows any text. When using this parameter, consider adding geographical information on where this is law.
| Legal status | |
|---|---|
| Legal status |
|
| legal_AU = S2
| legal_AU_comment = any text
| legal_BR = A1
| legal_BR_comment = any text
| legal_CA =
| legal_DE =
| legal_UK = gsl
| legal_US = Schedule II
| legal_US_comment = and OTC in Oregon
| legal_EU =
| legal_UN =
| legal_NZ =
| legal_status = Not marketed in Asia
Note: if a drug is restricted everywhere to prescription-only, please set legal_status = Rx-only rather than similarly define for each and every country. |legal_status=Rx-only is specifically recognised and shows as
- In general: ℞ (Prescription only)
| Input | Meaning |
|---|---|
| legal_status | Anywhere |
| Rx-only | ℞ Prescription only |
| legal_AU | Australia (see SUSMP) |
| Unscheduled | Unscheduled/exempt |
| S2 | Schedule 2 Pharmacy Medicine |
| S3 | Schedule 3 Pharmacist Only Medicine |
| S4 | Schedule 4 Prescription Only Medicine |
| S5 | Schedule 5 Caution |
| S6 | Schedule 6 Poison |
| S7 | Schedule 7 Dangerous Poison |
| S8 | Schedule 8 Controlled Drug |
| S9 | Schedule 9 Prohibited Substance |
| legal_BR | Brazil (see Controlled Drugs and Substances Act) |
| OTC | Over the counter |
| A1 | Class A1: Narcotic Drugs |
| A2 | Class A2: Narcotic Drugs |
| A3 | Class A3: Psychoactive Drugs |
| B1 | Class B1: Psychoactive Drugs |
| B2 | Class B2: Anorectic Drugs |
| C1 | Class C1: Other controlled substances |
| C2 | Class C2: Retinoids |
| C3 | Class C3: Immunosuppressive Drugs |
| C4 | |
| C5 | Class C5: Anabolic steroids |
| D1 | Class D1: Drug precursors |
| D2 | Class D2: Drug precursors |
| E | Class E: Controlled plants |
| F1 | Class F1: Prohibited narcotics |
| F2 | Class F2: Prohibited psychotropics |
| F3 | Class F3: Prohibited drug precursors |
| F4 | Class F4: Prohibited drug precursors |
| legal_CA | Canada |
| OTC | Over the counter |
| Rx-only | ℞ Prescription only |
| Schedule I | See Controlled Drugs and Substances Act |
| Schedule II | |
| Schedule III | |
| Schedule IV | |
| Schedule V | |
| Schedule VI | |
| Schedule VII | |
| Schedule VIII | |
| legal_UK | United Kingdom |
| GSL | General Sale List |
| P | Pharmacy Medicine |
| POM | Prescription Only Medicine |
| CD | Controlled Drug; if known may specify: |
| CD Lic | See Misuse of Drugs Regulations 2001 (as amended) |
| CD POM | |
| CD No Reg POM | |
| CD (Benz) POM | |
| CD (Anab) POM | |
| CD Inv POM | |
| legal_US | United States |
| OTC | Over the counter |
| ℞-only Rx-only |
Prescription only; if appropriate may specify: |
| Schedule I | See Controlled Substances Act |
| Schedule II | |
| Schedule III | |
| Schedule IV | |
| Schedule V | |
| EU | European Union |
| UN | United Nations |
| NZ | New Zealand |
Pharmacokinetic data
[edit]Use wikilinks for values that the general reader might not understand (e. g. hepatic, CYP3A4, intraperitoneal).
| bioavailability =
| protein_bound =
| metabolism =
| metabolites =
| onset =
| elimination_half-life =
| duration_of_action=
| excretion =
Names and identifiers
[edit]| parameter | label | demo | order | group | note | |
|---|---|---|---|---|---|---|
| {{PAGENAME}} | Amoxicillin | top | title | Infobox title | ||
drug_name =
|
N,N-Dimethyltryptamine | top | title | demo:1; overwrites {{PAGENAME}} | ||
| (type=vacc,mab,comb,other) | 01 | type | See /doc/type-sections | |||
IUPAC_name = |
Systematic (IUPAC) name | (2S,5R,6R)-6-{[(2R)-2-aminoetc. |
02 | type | Plain, long | |
SMILES = orsmiles =
|
SMILES | O=C(O)[C@@H]2N3C(=O)[C@@H]etc. |
03 | type | Plain, long, collapses | |
StdInChI =StdInChIKey = |
InChI | InChI=1S/C16H19N3O5S/c1-16(2)11etc.
Key:LSQZJLSetc. |
04 | type | Long, collapses. Not InChI, InChIKey | |
| Clinical data | 20 | clinical | ||||
tradename = |
Trade names | Actimoxi, Alphamox, etc. | 21 | clinical | Plain demo | |
Drugs.com = |
AHFS/Drugs.com | Monograph | 23 | clinical | Input by {{drugs.com}} | |
MedlinePlus = |
MedlinePlus | a685001 | 25 | clinical | ||
licence_EU =DailyMedID =licence_US = |
Licence data |
|
26 | clinical | DailyMedID overwrites licence_US | |
| Identifiers | 50 | id | ||||
CAS_number =CAS_supplemental = |
CAS Registry Number | 91161-71-6 78628-80-5 | 51 | id | demo:4 | |
ATC_prefix = ATC_suffix =ATC_supplement = ATCvet = |
ATC code or ATCvet code |
ATCvet=no |ATC_prefix=J01 |ATC_suffix=CA04 |ATC_supplemental=QG51AA03 (WHO) }} | 52 | class | Multi-input, interaction. Numbers have split wl+el! | |
PubChem = orPubChemSubstance = (SID) |
PubChem | CID: 33613 | 53 | id | Only one is shown | |
IUPHAR_ligand =
|
IUPHAR/BPS | 4139 | 54 | id | demo:2 | |
DrugBank =
|
DrugBank | DB01060 | 55 | id | ||
ChemSpiderID =
|
ChemSpider | 31006 | 56 | id | +Option '=none' | |
UNII =
|
UNII | 9EM05410Q9 | 57 | id | ||
KEGG =
|
KEGG | D07452 | 58 | id | ||
ChEBI =
|
ChEBI | CHEBI:2676 | 59 | id | ||
ChEMBL =
|
ChEMBL | CHEMBL1082 | 61 | id | ||
NIAID_ChemDB =
|
NIAID ChemDB | 059486 | 62 | id | demo:3; HIV/AIDS related | |
synonyms =
|
Synonyms | 2-acetoxybenzoic acid acetylsalicylate etc. |
63 | id | demo:2; Plain, list | |
PDB_ligand =
|
PDB ligand ID | AIN (PDBe, RCSB PDB) | 64 | id | demo:2 | |
E_number =
|
E number | E703 | 65 | id | Oxytetracycline | |
- Notes: Data is taken from various articles, for illustration purposes.
AAN, BAN, JAN, USAN
[edit]Some countries have a variant INN name defined. For example, USAN (US) uses "acetaminophen" for paracetamol. Use |AAN= BAN= JAN= USAN= when a name is different from the INN (do not repeat INN).
CAS Registry Number
[edit]|CAS_number=
Additional CAS codes may be included with optional |CAS_supplement= parameters in a comma-separated lists. CAS templates may be used to have these additional codes link to the relevant databases:
| CAS_supplemental ={{CAS|427-51-0}} (acetate)
Adds the following linked codes:
- For CAS – 427-51-0 (acetate)
- See also: Second identifiers and indexes
DrugBank
[edit]The DrugBank primary accession number (consisting of a 2 letter prefix (DB) and a 5 number suffix). Secondary accession numbers with a 4 letter suffix (APRD, EXPT, BIOD, NUTR) should not normally be used.
PubChem
[edit]When available, the PubChem compound identifier (CID) should be used because it is unique for each chemical compound:
| PubChem = 4091 <!-- Metformin -->
Make sure you choose the right CID: Often PubChem compound entries differ only very slightly, for example by an additional hydratation water or by a carbon atom with unspecified stereochemistry.
If no CID is available, which is usually the case when there is no structural information on PubChem, you may use one of the substance identifiers (SIDs):
| PubChemSubstance = 10099 <!-- Etanercept -->
CompTox (EPA database)
[edit]|DTXSID= is the identifier for the CompTox Chemicals Dashboard (by EPA). For example: |DTXSID=DTXSID5020108 for aspirin.
By default, the value is read from Wikidata DSSTox substance ID (P3117). This can be overwritten by entering a value for |DTXSID=. Setting |DTXSID=none suppresses the Wikidata value (no show).
Second identifier (like: CAS_number2)
[edit]
Using second identifiers
| ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
All identifiers can have a second, index-2 identifier, representing a second chemical substance. For example, Additionally,
{{infobox drug
| drug_name=Demo indexes<br/>([[Diminazen]])
<!-- Identifiers -->
| index_label=
| index2_label=aceturate
| index_comment = standard
| index2_comment = see [[aceturic acid]]
| CAS_number = 536-71-0
| CAS_number2 = 908-54-3
| PubChem = 2354
| UNII = Y5G36EEA5Z
| UNII2 = JI8SAD85NO
| DrugBank =
}}
| ||||||||||||||||||||||||||||
Chemical and physical data
[edit]Chemical formula
[edit]Entering the chemical formula per element:
| C= | H= | Ag= | Al= | As= | Au= | B= | Bi= | Br= | Ca= | Cl= | Co= | F= | Fe= | Gd= | I=
| K= | Li= | Mg= | Mn= | N= | Na= | O= | P= | Pt= | S= | Sb= | Se= | Sr= | Tc= | Zn=
<!-- all 118 symbols -->
| charge=
| chemical_formula_ref =
| chemical_formula_comment =
E.g.
| C=2 | H=6 | O=1 | charge=2-
gives
- Formula C2H6O2−
This is called the empirical form, the most simple form. Entered this way, the elements are ordered in the Hill notation order: When C is present, CxHy in front and the others alphabetically Ar ... Zr. When a molecular formula or structural formula is known, that one should be entered in |chemical_formula= (consider using {{Chem2}}).
You can provide the chemical formula as fixed
| chemical_formula =
| chemical_formula_ref =
| chemical_formula_comment =
todo: example
- Molar mass
When the chemical formula is entered as |C=2 |O=1 |H=6, the molar mass is calculated and presented. This value can be overwritten by using |molecular_weight= (sic). For more information, see {{Chem molar mass}}.
Other chemical data
[edit]Additional chemical data fields are SMILES and standard InChI (optionally oncluding a comment and standard InChIKey).
| SMILES =
| Jmol =
| StdInChI =
| StdInChI_comment =
| StdInChIKey =
- The Jmol 3D model
When |SMILES= has input, the template automatically adds an external link to the Jmol 3D molecule model.
|Jmol=nonewill suppress (hide) that data row ("none" is case-sensitive).|Jmol=⟨some SMILES string⟩will link to the 3D-model of that string (i.e. overwriting|SMILES=input). SMILES will show its input unchanged.
Physical data
[edit]This is entirely optional data, and for most drugs is not helpful to the wider readership. Only include if information of particular interest for the drug as to its chemical properties (e.g. in its manufacture or as an important chemical in its own right, e.g. Aspirin).
| density =
| density_notes =
| melting_point =
| boiling_point =
| solubility =
| sol_units =
| specific_rotation =
The template will add the following to the numeric values provided:
- Density – added 'g/cm3'
- Melting or Boiling points – added '°C' along with calculated converted value in °F.
- Solubility – If sol_units is specified, it will follow the solubility value; otherwise " mg/mL (20 °C)" will follow. This is to accommodate multiple solubility values at different temps, other units, etc.
Hence:
| solubility = 100
| sol_units = &nbsp;g/L ({{Convert|212|F|C}})
gives:
- Solubility 100 g/L (212 °F (100 °C))
In addition, where the melting point occurs over a range of temperatures, use melting_high for the upper value.
Hence:
| melting_point = 100
| melting_high = 104
gives:
- Melting point 100–104 °C (212–219 °F)
Comments can be added to the melting and boiling point entries using melting_notes and boiling_notes.
Hence:
| boiling_point = 100
| boiling_notes = (sublimes)
gives:
- Boiling point 100 °C (212 °F) (sublimes)
Physiological data (endogenous drugs)
[edit]Endogenous drugs (neurotransmitters, neurohormones, or hormones) are a single chemical (|type=<blank>). Their special data is shown in section "Physiological data".
Notes: input |metabolism= is shared with pharmacokinetic data, and will show in each of these sections that has more data (context).
When this section has input, sections Clinical data and Legal data should be empty.
<!-- Physiological data -->
| source_tissues =
| target_tissues =
| receptors =
| agonists =
| antagonists =
| precursor =
| biosynthesis =
| metabolism = <!-- same parameter as in pharmacokinetic data -->
Example:
| drug_name = [[Oxytocin]]
<!-- Physiological data -->
| source_tissues = [[posterior pituitary]]
| target_tissues = [[central nervous system]]
| receptors = [[oxytocin receptor]]
| agonists = [[carbetocin]], [[demoxytocin]]
| antagonists = [[atosiban]], [[epelsiban]]
| precursor = [[Neurophysin I|oxytocin-neurophysin 1]]
| biosynthesis = [[magnolysin]]
| metabolism = [[oxytocinase]]
- Categorised: Category:Drugs that are a physiological drug (21)
Gene therapy
[edit]| gt_target_gene =
| gt_vector =
| gt_nucleic_acid_type =
| gt_editing_method =
| gt_delivery_method =
Example:
| Gene therapy | |
|---|---|
| Target gene | RPE65 |
| Vector | adenovirus serotype 2 |
| Nucleic acid type | [not applicable here] |
| Editing method | [not applicable here] |
| Delivery method | [not applicable here] |
{{Infobox drug
| drug_name = [[Voretigene neparvovec]]
| gt_target_gene = [[RPE65]]
| gt_vector = [[Adeno-associated virus|adenovirus serotype 2]]
| gt_nucleic_acid_type = [not applicable here]
| gt_editing_method = [not applicable here]
| gt_delivery_method = [not applicable here]
}}
- Categorised: Category:Drugs that are a gene therapy (14)
Input from Wikidata
[edit] E number (P628) (see uses)
E number (P628) (see uses) ECHA Substance Infocard ID (P2566) (see uses)
ECHA Substance Infocard ID (P2566) (see uses) DSSTox substance ID (P3117) (see uses)
DSSTox substance ID (P3117) (see uses)
The template reads and shows the E number, ECHA InfoCard ID and Comptox DTXSID ID from Wikidata. If there is no value present, no data is shown.
See also Category:Chemical compounds and Wikidata.
Miscellaneous
[edit]Verification by CheMoBot (parameters: ..._Ref)
[edit]Seven identifying parameters like |CAS_number= are tracked in a WP:Drugbox data validation process. A bot adds parameters like
|verifiedrevid= and |CAS_number_Ref={{cascite|correct}}. See also {{cascite}} documentation.
English variant spellings (ENGVAR)
[edit]The word "License/Licence" can be spelled in two ways, generally by English variant en-UK or en-US. Per Manual of Style:ENGVAR, this chosen regional language should be consistent throughout the whole article.
The default spelling in the infobox is en-US: "LicenSe". Setting |engvar=en-AU, en-CA, en-EI, en-NZ, en-UK spells "LicenCe". Again, this should follow the article's overall language.
Independently from this, each parameter |licence_US= or |license_US= can be used for the same input point. This is for ease of editing, but does not change the spelling of "Licence/License" that will show.
Section overview by |type= options
[edit]A |type= can use specific headers.
| type=mab
| type=vaccine
| type=combo
| type=
- Types are listed in Category:Drugs by type
Dependent sections:
Infobox drug sections
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Multiple Infoboxes drug
[edit]- Eprinomectin, Hib vaccine, Sarafotoxin (3 boxes), Valproate, Varespladib
- (checked manually using WP:AWB, 5/8275, 20-12-2021)
TemplateData
[edit]|
See a monthly parameter usage report for Template:Infobox drug in articles based on its TemplateData.
TemplateData for Infobox drug This infobox provides information over-the-counter and prescription-only drugs. It can be used for single drugs, combination products, monoclonal antibodies and vaccines.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Tracking categories
[edit]- Category:Pages using infobox drug with unknown parameters (9)
- See also: Category:Infobox drug tracking categories
See also
[edit]- {{Infobox drug class}}
- {{Infobox medical condition}}
- WP:PHARM for WikiProject page
- Wikipedia Drugbox and Chembox Maker

