Semidione
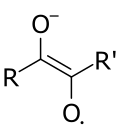
Semidiones are radical anions analogous to semiquinones, obtained from the one-electron reduction of non-quinone conjugated dicarbonyls.[1]
The simplest possible semidiones are derived from 1,2-dicarbonyls and have structure R−C(−O−)=C(−O•)−R' ↔ R−C(−O•)=C(−O−)−R', making them the second member of a homologous series starting with ketyl radicals and continuing with semitriones.[1] They are often transient intermediates, appearing in reactions such as the final reduction step of the acyloin condensation.[2]
Benzil semidione (Ph−C(−O−)=C(−O•)−Ph ↔ Ph−C(−O•)=C(−O−)−Ph), synthesized by Auguste Laurent in 1836, is believed to have been the first radical ion ever characterized.[3]: 425 [4]
Semidehydroascorbate is a relatively stable semitrione produced by hydrogen abstraction from ascorbate (Vitamin C).[5]
References
[edit]- ^ a b Russell, Glen A. (1968). "Chapter 3: Semidione Radical Anions". In Kevan, Larry; Kaiser, Emil T. (eds.). Radical Ions. New York Interscience. pp. 87–150. ISBN 978-0-470-45490-9.
- ^ Roth, Heinz D. (2004). "Chapter 6: Organic Radical Ions". In Moss, Robert A.; Platz, Matthew; Jones, Maitland (eds.). Reactive Intermediate Chemistry. Wiley-Interscience. p. 260. ISBN 978-0-471-23324-4.
- ^ Russell, Glen A.; Norris, Robert K. (1973). "Chapter 6: Radical Ions". In McManus, Samuel P. (ed.). Organic Reactive Intermediates. Organic Chemistry. Vol. 26. Elsevier. pp. 423–448. doi:10.1016/B978-0-12-485450-5.50011-7. ISSN 0078-611X.
- ^ Laurent, Auguste (1836), "Benzoyl und Benzimid.", Annalen der Pharmacie, 17 (1): 88–94, doi:10.1002/jlac.18360170116, hdl:2027/mdp.39015026322357
- ^ Njus, David; Kelley, Patrick M.; Tu, Yi-Jung; Schlegel, H. Bernhard (November 2020). "Ascorbic acid: The chemistry underlying its antioxidant properties". Free Radical Biology and Medicine. 159: 37–43. doi:10.1016/j.freeradbiomed.2020.07.013.
