Charcot–Marie–Tooth disease
| Charcot–Marie–Tooth disease | |
|---|---|
| Other names | Charcot–Marie–Tooth neuropathy, peroneal muscular atrophy, Dejerine-Sottas syndrome |
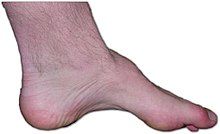 | |
| The foot of a person with Charcot–Marie–Tooth disease: The lack of muscle, a high arch, and claw toes are signs of this genetic disease. | |
| Pronunciation | |
| Specialty | Neurology, podiatry, orthopedics, physical medicine and rehabilitation |
| Symptoms | Common: high-arched feet, hammertoe, foot drop, high-stepping gait, weakness, stiffness, and muscle wasting of lower legs, arm, and hands, and reduced tendon reflexes. Sometimes: flat-arched feet, spinal deformities.[1][2] |
| Usual onset | Childhood – early adulthood |
| Duration | Lifelong |
| Causes | Family history (genetics) |
| Risk factors | Family history (genetics) |
| Diagnostic method | Genetic testing, nerve conduction study or electromyogram (EMG) |
| Differential diagnosis | Muscular dystrophy |
| Treatment | Management to maintain function |
| Prognosis | Progressive |
| Frequency | Prevalence: 1 in 2,500[3][4] |
Charcot–Marie–Tooth disease (CMT) is a hereditary motor and sensory neuropathy of the peripheral nervous system characterized by progressive loss of muscle tissue and touch sensation across various parts of the body. This disease is the most commonly inherited neurological disorder, affecting about one in 2,500 people.[5][6] It is named after those who classically described it: the Frenchman Jean-Martin Charcot (1825–1893), his pupil Pierre Marie (1853–1940),[7] and the Briton Howard Henry Tooth (1856–1925).[8][9]
There is no known cure. Care focuses on maintaining function. CMT was previously classified as a subtype of muscular dystrophy.[5]
Signs and symptoms
[edit]Symptoms of CMT usually begin in early childhood or early adulthood but can begin later. Some people do not experience symptoms until their early 30s or 40s. Usually, the initial symptom is foot drop or high arches early in the course of the disease. This can be accompanied by hammertoe, where the toes are always curled. Wasting atrophy of muscle tissue of the lower parts of the legs may give rise to a "stork leg" or "inverted champagne bottle" appearance. Weakness in the hands and forearms occurs in many people as the disease progresses.[10]
High-arched feet (pes cavus) or flat-arched feet (pes planus) are classically associated with the disorder.[11] Loss of touch sensation in the feet, ankles, and legs as well as in the hands, wrists, and arms occurs with various types of the disease. Early- and late-onset forms occur with 'on and off' painful spasmodic muscular contractions that can be disabling when the disease activates. Sensory and proprioceptive nerves in the hands and feet are often damaged, while unmyelinated pain nerves are left intact. Overuse of an affected hand or limb can activate symptoms including numbness, spasm, and painful cramping.[10]
Symptoms and progression of the disease can vary. Involuntary grinding of teeth and squinting are prevalent and often go unnoticed by the person affected. Breathing can be affected in some, as can hearing, vision, and neck and shoulder muscles. Scoliosis is common, causing hunching and loss of height. Hip sockets can be malformed. Gastrointestinal problems can be part of CMT,[12][13] as can difficulty chewing, swallowing, and speaking (due to atrophy of vocal cords).[14] A tremor can develop as muscles waste. Pregnancy has been known to exacerbate CMT, as well as severe emotional stress. Patients with CMT must avoid periods of prolonged immobility such as when recovering from a secondary injury, as prolonged periods of limited mobility can drastically accelerate symptoms of CMT.[15]
Pain due to postural changes, skeletal deformations, muscle fatigue, and cramping is fairly common in people with CMT. It can be mitigated or treated by physical therapies, surgeries, and corrective or assistive devices. Analgesic medications may also be needed if other therapies do not provide relief from pain.[16] Neuropathic pain is often a symptom of CMT, though, like other symptoms of CMT, its presence and severity vary from case to case. For some people, pain can be significant to severe and interfere with daily life activities. However, pain is not experienced by all people with CMT. When neuropathic pain is present as a symptom of CMT, it is comparable to that seen in other peripheral neuropathies, as well as postherpetic neuralgia and complex regional pain syndrome, among other diseases.[17]
Atypical presentations of CMT can also lead to leg muscles, specifically the calves, enlarging.[18] This hypertrophic type of CMT is not caused by the muscles enlarging directly, but by pseudohypertrophy of the legs as fatty tissue enters the leg muscles.[19][20][21]
Causes
[edit]
Charcot–Marie–Tooth disease is caused by genetic mutations that cause defects in neuronal proteins. Nerve signals are conducted by an axon with a myelin sheath wrapped around it. Most mutations in CMT affect the myelin sheath, but some affect the axon.[22]
Chromosome 17
The most common cause of CMT (70–80% of the cases) is the duplication of a large region on the short arm of chromosome 17 that includes the gene PMP22.[23]
Chromosome 1
Some mutations affect the gene MFN2, on chromosome 1, which codes for a mitochondrial protein. Mutated MFN2 causes the mitochondria to form large clusters, or clots, which are unable to travel down the axon towards the synapses. This prevents the synapses from functioning.[24]
Classification
[edit]CMT is a heterogeneous disease and the mutations linked to it may occur in many different genes.[25] Based on the affected gene, CMT is categorized into several types and subtypes.[26]
GARS1-Related Axonal Neuropathy (CMT2)
[edit]CMT2 variants are typically referred to as axonal neuropathies due to the axonal degeneration observed. CMT2 variants are a result of damage to the nerve axons, rather than damage to the myelin sheath (as is the case with CMT1). Damaged axons cause slowed transmission of signals to the muscles and brain, causing symptoms including muscle atrophy, weakness, decreased sensitivity, and foot deformity. Symptoms of CMT2 variants typically appear between the ages of 5 and 25.[27] CMT2D is one of 31 CMT2 variants, and is only diagnosed if sensory deficits (such as loss of sensation due to the degradation of sensory axons) are observed along with motor deficits; otherwise, distal hereditary motor neuropathy type V is diagnosed. It is unknown why sensory involvement is so varied between GARS1 neuropathy patients.[28] Symptoms of CMT2D include foot deformity, muscle weakness and cramping, compromised reflexes, loss of sensation, and muscle atrophy, and are similar to the symptoms of both CMT1 and CMT2 variants. Symptoms and severity vary from patient to patient.[29]
Mice are often used to model CMT2D, and typically demonstrate aberrant neuromuscular function at the neuromuscular junction (NMJ).[30][31][32] The neuromuscular junction is abnormal in CMT2D mice, with subjects showing neuromuscular junction degeneration in hind muscles. The dorsal root ganglia (DRG) are also affected via aberrant sensory neuron fate, meaning that sensory neuron cell fates are abnormally determined. CMT2D mice have fewer proprioceptive and mechanosensitive neurons, but have more nociceptive neurons, possibly due to mutant GlyRS aberrantly interacting with the extracellular region of tropomyosin receptor kinase, or Trk, receptors.[33] Trk receptors are crucial to the survival and development of sensory neurons; when disrupted, nerve development and survival is disrupted as well, possibly leading to the abnormal sensory neuron counts observed in CMT2D mice.[28]
CMT2D is a result of autosomal dominant mutations in the human GARS1 gene located at 7p14.3 [34] and is thought to be caused by aberrant gain-of-function missense mutations.[28] The GARS1 gene is a protein-coding gene responsible for the encoding of glycyl-tRNA synthetase (GlyRS). Glycyl-tRNA synthetase is a class II aminoacyl-tRNA synthetase and acts as the catalyst for the synthesis of glycyl-tRNA by covalently bonding amino acids with their corresponding cognate tRNAs for protein translation. Glycyl-tRNA synthetase is integral to protein translation and attaches glycine to its cognate tRNA.[35]
Many different mutations have been found in CMT2D patients, and it remains unclear how mutations in GARS1 cause CMT2D. However, it is thought that mutant glycyl-tRNA synthetase (GlyRS) interferes with transmembrane receptors, causing motor disease,[36][37] and that mutations in the gene could disrupt the ability of GlyRS to interact with its cognate RNA, disrupting protein production. The GARS1 mutations present in CMT2D cause a deficient amount of glycyl-tRNA in cells, preventing the elongation phase of protein synthesis. Elongation is a key step in protein production, so when there is a deficiency of glycyl-tRNA, protein synthesis is unable to continue at glycine sites. GARS1 mutations also stall initiation of translation due to a stress response that is induced by glycine addition failure. By stalling elongation and initiation of translation, CMT2D mutations in the GARS1 gene cause translational repression, meaning that overall translation is inhibited.[38]
GARS1-associated axonal neuropathy is progressive, meaning that it worsens over time. Unknown mechanisms are thought to cause the chronic neurodegeneration resulting from the aberrant GlyRS; however, one theory on the mechanism for the disease is VEGF deficiency. Mutant GlysRS interferes with neuronal transmembrane receptors, including neuropilin 1 (Nrp1) and vascular endothelial growth factor (VEGF), causing neuropathy.[37] GARS-CMT2D mutations alter GlyRS and allow it to bind to the Nrp1 receptor, interfering with the normal binding of Nrp1 to VEGF. While enhanced expression of VEGF improves motor function, reduced expression of Nrp1 worsens CMT2D; because Nrp1 binds to mutant GlyRS in mutant GARS1-CMT2D individuals, Nrp1 expression is reduced, in turn worsening motor function. Mice with deficient VEGF demonstrate motor neuron disease over time. Thus, the VEGF/Nrp1 pathway is considered to be targetable for CMT2D treatment.[27]
X-linked CMT
[edit]CMT can also be produced by X-linked mutations, in which case it is called X-linked CMT (CMTX). In CMTX, mutated connexons create nonfunctional gap junctions that interrupt molecular exchange and signal transport.[39][40][41] The mutation can appear in the GJB1 gene coding for the connexin 32 protein, a gap junction protein expressed in Schwann cells. Because this protein is also present in oligodendrocytes, demyelination can appear in the CNS as well.[42]
Schwann cells create the myelin sheath by wrapping their plasma membranes around the axon.[39] These Schwann cells work together with neurons and fibroblasts to create a functional nerve. Schwann cells and neurons exchange molecular signals by way of gap junctions that regulate survival and differentiation[43]
Demyelinating Schwann cells cause abnormal axon structure and function. They may cause axon degeneration, or they may simply cause axons to malfunction.[5] The myelin sheath allows nerve cells to conduct signals faster. When the myelin sheath is damaged, however, nerve signals are slower. This can be measured by a common neurological test, electromyography. When the axon is damaged, the result is a reduced compound muscle action potential.[44]
Diagnosis
[edit]CMT can be diagnosed through three different forms of tests: measurement of the speed of nerve impulses (nerve conduction studies), a biopsy of the nerve, and DNA testing. DNA testing can give a definitive diagnosis, but not all the genetic markers for CMT are known. CMT is first most noticed when someone develops lower leg weakness, such as foot drop, or foot deformities, including hammertoes and high arches, but signs alone do not lead to diagnosis. Patients must be referred to a physician specialising in neurology or rehabilitation medicine. To see signs of muscle weakness, the neurologist may ask patients to walk on their heels or to move part of their leg against an opposing force. To identify sensory loss, the neurologist tests for deep-tendon reflexes, such as the knee jerk, which are reduced or absent in CMT. The doctor may also ask about the patient's family history since CMT is hereditary. The lack of family history does not rule out CMT but is helpful to rule out other causes of neuropathy, such as diabetes or exposure to certain chemicals or drugs.[45]
In 2010, CMT was one of the first diseases where the genetic cause of a particular patient's disease was precisely determined by sequencing the whole genome of an affected individual. This was done by the scientists employed by the Charcot Marie Tooth Association (CMTA).[46][26] Two mutations were identified in a gene, SH3TC2, known to cause CMT. Researchers then compared the affected patient's genome to the genomes of the patient's mother, father, and seven siblings with and without the disease. The mother and father each had one normal and one mutant copy of this gene and had mild or no symptoms. The offspring who inherited two mutant genes presented fully with the disease.[26]
Histology
[edit]
The constant cycle of demyelination and remyelination, which occurs in CMT, can lead to the formation of layers of myelin around some nerves, termed an "onion bulb". These are also seen in chronic inflammatory demyelinating polyneuropathy.[47] Muscles show fiber type grouping, a similarly nonspecific finding that indicates a cycle of denervation/reinnervation. Normally, type I and type II muscle fibers show a checkerboard-like random distribution. However, when reinnervation occurs, the group of fibers associated with one nerve are of the same type. The standard for indicating fiber type is histoenzymatic adenosine triphosphatase (ATPase at pH 9.4).[48]
Management
[edit]Often, the most important goal for patients with CMT is to maintain movement, muscle strength, and flexibility. Therefore, an interprofessional team approach with occupational therapy (OT), physical therapy (PT), orthotist, podiatrist, and or orthopedic surgeon is recommended.[10]
Appropriate footwear is also very important for people with CMT, but they often have difficulty finding well-fitting shoes because of their high-arched feet and hammertoes. Due to the lack of good sensory reception in the feet, CMT patients may also need to see a podiatrist for assistance in trimming nails or removing calluses that develop on the pads of the feet. Lastly, patients can also decide to have surgery performed by a podiatrist or an orthopedic surgeon. Surgery may help to stabilize the patients' feet or correct progressive problems. These procedures include straightening and pinning the toes, lowering the arch, and sometimes, fusing the ankle joint to provide stability.[15] CMT patients must take extra care to avoid falling as fractures take longer to heal in someone with an underlying disease process. Additionally, the resulting inactivity may cause the CMT to worsen.[15] The Charcot–Marie–Tooth Association classifies the chemotherapy drug vincristine as a "definite high risk" and states, "vincristine has been proven hazardous and should be avoided by all CMT patients, including those with no symptoms."[49] Several corrective surgical procedures can be done to improve the physical condition of the affected individuals.[50]
Orthotics
[edit]
If the muscles of the lower extremities are weak, it makes sense to prescribe custom-fabricated orthotics. Depending on which muscle groups are affected, the correct orthoses with appropriate functional elements should be prescribed. A weakness of the tibialis anterior muscle, which lifts the feet, is usually accompanied by an atrophy of the gastrocnemius muscle which, together with the soleus muscle, forms the triceps surae muscles (distal calf muscles), occurs causing the known "stork leg deformity".[51] In most cases, ankle-foot orthoses that have functional elements for the foot lifting and adjustable control of the lowering of the forefoot make sense. Weak calf muscles lead to insufficient activation of the forefoot lever. This leads to an additional increasing uncertainty when standing and walking. If the calf muscles are weak, an orthosis should therefore be equipped with functional elements to activate the forefoot lever. An orthotic joint with an adjustable dynamic dorsiflexion stop with a strong spring in combination with a lower leg shell in front of the shin is recommended for this. Such orthoses help to control foot drop, and instability of the foot and ankle and offer the patient a better sense of balance when standing and walking without restricting mobility and the dynamics of the ankle joint. Studies confirm the positive effect of orthoses with adjustable functional elements in patients with paralysis of these muscle groups.[52][53][54][55] It is of great advantage if the resistances of the two functional elements can be set separately from one another in the two directions of movement, dorsiflexion and plantar flexion.[56]
Prognosis
[edit]The severity of symptoms varies widely even for the same type of CMT. Cases of monozygotic twins with varying levels of disease severity have been reported, showing that identical genotypes are associated with different levels of severity (see penetrance). Some patients can live a normal life and are almost or entirely asymptomatic.[57] A 2007 review stated that "life expectancy is not known to be altered in the majority of cases."[58]
History
[edit]The disease is named after those who classically described it: the Frenchman Jean-Martin Charcot (1825–1893), his pupil Pierre Marie (1853–1940),[7] and the Briton Howard Henry Tooth (1856–1925).[8]
See also
[edit]- Charcot–Marie–Tooth disease classifications
- Palmoplantar keratoderma and spastic paraplegia
- Hereditary motor and sensory neuropathies
- Hereditary motor neuropathies
- Low copy repeats
- Christina's World
References
[edit]- ^ Szigeti K, Lupski JR (2009). "Charcot–Marie–Tooth disease". European Journal of Human Genetics. 17 (6): 703–710. doi:10.1038/ejhg.2009.31. PMC 2947101. PMID 19277060.
- ^ Nagappa M, Sharma S, Taly AB (2024), "Charcot-Marie-Tooth Disease", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 32965834, retrieved 2024-09-20
- ^ Cornett KM, Menezes MP, Bray P, Halaki M, Shy RR, Yum SW, et al. (June 2016). "Phenotypic Variability of Childhood Charcot-Marie-Tooth Disease". JAMA Neurology. 73 (6): 645–651. doi:10.1001/jamaneurol.2016.0171. PMC 4916861. PMID 27043305.
- ^ Skre H (August 1974). "Genetic and clinical aspects of Charcot-Marie-Tooth's disease". Clinical Genetics. 6 (2): 98–118. doi:10.1111/j.1399-0004.1974.tb00638.x. PMID 4430158.
- ^ a b c Krajewski KM, Lewis RA, Fuerst DR, Turansky C, Hinderer SR, Garbern J, et al. (July 2000). "Neurological dysfunction and axonal degeneration in Charcot-Marie-Tooth disease type 1A". Brain. 123 (7): 1516–1527. doi:10.1093/brain/123.7.1516. PMID 10869062.
- ^ Physical Medicine and Rehabilitation for Charcot-Marie-Tooth Disease at eMedicine
- ^ a b Charcot JM (1886). "Sur une forme particulière d'atrophie musculaire progressive, souvent familiale débutant par les pieds et les jambes et atteignant plus tard les mains" [On a particular form of progressive muscular atrophy, often familial, beginning with the feet and legs and later reaching the hands]. Revue Médicale (in French). 6: 97–138.
- ^ a b Tooth HH (1886). The peroneal type of progressive muscular atrophy (MD thesis). London.
- ^ Kumar DR, Aslinia F, Yale SH, Mazza JJ (March 2011). "Jean-Martin Charcot: The Father of Neurology". Clinical Medicine & Research. 9 (1): 46–49. doi:10.3121/cmr.2009.883. PMC 3064755. PMID 20739583.
- ^ a b c "Charcot-Marie-Tooth Disease Fact Sheet". National Institute of Neurological Disorders and Stroke. Retrieved 2022-04-23.
- ^ Le T, Bhushan V (2013). First Aid for the USMLE Step 1 2014. McGraw-Hill Education. ISBN 978-0-07-183142-0.
Typically autosomal dominant inheritance pattern associated with scoliosis and foot deformities (high or flat arches).
[page needed] - ^ "CMT News". Lindacrabtree.com. Archived from the original on 2016-08-05. Retrieved 2016-11-13.
- ^ Soykan I, McCallum RW (January 1997). "Gastrointestinal involvement in neurologic disorders: Stiff-man and Charcot-Marie-Tooth syndromes". The American Journal of the Medical Sciences. 313 (1): 70–73. doi:10.1097/00000441-199701000-00012. PMID 9001170.
- ^ "Charcot-Marie-Tooth Disease Fact Sheet". National Institute of Neurological Disorders and Stroke. 2016-01-14. Archived from the original on 2016-11-19. Retrieved 2016-11-13.
- ^ a b c "Treatment and Management of CMT" (Press release). Charcot-Marie-Tooth Association. October 6, 2010. Retrieved August 26, 2011.
- ^ "Charcot-Marie-Tooth Syndrome. CMT information". Patient. 20 August 2021.
- ^ Carter GT, Jensen MP, Galer BS, Kraft GH, Crabtree LD, Beardsley RM, et al. (December 1998). "Neuropathic pain in Charcot-Marie-Tooth disease". Archives of Physical Medicine and Rehabilitation. 79 (12): 1560–1564. doi:10.1016/S0003-9993(98)90421-X. PMID 9862301.
- ^ Nan H, Wu Y, Cui S, Sun H, Wang J, Li Y, et al. (2022). "Coexistence of Charcot-Marie-Tooth 1A and nondystrophic myotonia due to PMP22 duplication and SCN4A pathogenic variants: a case report". BMC Neurology. 22 (1): 17. doi:10.1186/s12883-021-02538-5. PMC 8740465. PMID 34996390.
- ^ Krampitz DE, Wolfe GI, Fleckenstein JL, Barohn RJ (November 1998). "Charcot-Marie-Tooth disease type 1A presenting as calf hypertrophy and muscle cramps". Neurology. 51 (5): 1508–1509. doi:10.1212/WNL.51.5.1508. PMID 9818900.
- ^ Smith TW, Bhawan J, Keller RB, Degirolami U (July 1980). "Charcot-Marie-Tooth Disease Associated with Hypertrophic Neuropathy: A Neuropathologic Study of Two Cases". Journal of Neuropathology and Experimental Neurology. 39 (4): 420–440. doi:10.1097/00005072-198007000-00003. PMID 6260904.
- ^ Brusse E, Perumpillichira J (October 2014). "G.P.294". Neuromuscular Disorders. 24 (9–10): 910. doi:10.1016/j.nmd.2014.06.384.
- ^ Niemann A, Berger P, Suter U (March 2006). "Pathomechanisms of mutant proteins in Charcot-Marie-Tooth disease". NeuroMolecular Medicine. 8 (1–2): 217–241. doi:10.1385/nmm:8:1-2:217 (inactive 7 December 2024). hdl:20.500.11850/422903. PMID 16775378.
{{cite journal}}: CS1 maint: DOI inactive as of December 2024 (link) - ^ Florescu C, Albu CV, Dumitrescu C, Târtea GC, Florescu OA, Târtea EA (2017). "Sleep and Memory Disorders in a Patient Suffering from Charcot-Marie-Tooth Disease". Current Health Sciences Journal. 43 (1): 73–77. doi:10.12865/CHSJ.43.01.11. PMC 6286719. PMID 30595858.
- ^ Baloh RH, Schmidt RE, Pestronk A, Milbrandt J (January 2007). "Altered axonal mitochondrial transport in the pathogenesis of Charcot-Marie-Tooth disease from mitofusin 2 mutations". The Journal of Neuroscience. 27 (2): 422–430. doi:10.1523/JNEUROSCI.4798-06.2007. PMC 6672077. PMID 17215403.
- ^ Hoyle JC, Isfort MC, Roggenbuck J, Arnold WD (2015). "The genetics of Charcot-Marie-Tooth disease: current trends and future implications for diagnosis and management". The Application of Clinical Genetics. 8: 235–243. doi:10.2147/TACG.S69969. PMC 4621202. PMID 26527893.
- ^ a b c Lupski JR, Reid JG, Gonzaga-Jauregui C, Rio Deiros D, Chen DC, Nazareth L, et al. (April 2010). "Whole-genome sequencing in a patient with Charcot-Marie-Tooth neuropathy". The New England Journal of Medicine. 362 (13): 1181–1191. doi:10.1056/NEJMoa0908094. PMC 4036802. PMID 20220177.
- ^ a b "CMT2 - Types of Charcot-Marie-Tooth Disease (CMT) - Diseases". Muscular Dystrophy Association. 2015-12-23. Retrieved 2022-05-10.
- ^ a b c Sleigh JN, Mech AM, Aktar T, Zhang Y, Schiavo G (2020). "Altered Sensory Neuron Development in CMT2D Mice Is Site-Specific and Linked to Increased GlyRS Levels". Frontiers in Cellular Neuroscience. 14: 232. doi:10.3389/fncel.2020.00232. PMC 7431706. PMID 32848623.
- ^ "Charcot-Marie-Tooth disease type 2D (Concept Id: C1832274)". MedGen - NCBI. Retrieved 2022-05-10.
- ^ Sleigh JN, Grice SJ, Burgess RW, Talbot K, Cader MZ (2014). "Neuromuscular junction maturation defects precede impaired lower motor neuron connectivity in Charcot-Marie-Tooth type 2D mice". Hum Mol Genet. 15 (10): 2639–50. doi:10.1093/hmg/ddt659. PMC 3990164. PMID 24368416.
- ^ Spaulding EL, Sleigh JN, Morelli KH, Pinter MJ, Burgess RW, Seburn KL (2016). "Synaptic Deficits at Neuromuscular Junctions in Two Mouse Models of Charcot-Marie-Tooth Type 2d". J Neurosci. 16 (11): 3254–67. doi:10.1523/JNEUROSCI.1762-15.2016. PMC 4792937. PMID 26985035.
- ^ Sleigh JN, Mech AM, Schiavo G (2020). "Developmental demands contribute to early neuromuscular degeneration in CMT2D mice". Cell Death Dis. 11 (7): 564. doi:10.1038/s41419-020-02798-y. PMC 7378196. PMID 32703932.
- ^ Sleigh JN, Dawes JM, West SJ, Wei N, Spaulding EL, Gómez-Martín A, et al. (2017). "Trk receptor signaling and sensory neuron fate are perturbed in human neuropathy caused by Gars mutations". Proc Natl Acad Sci U S A. 114 (16): E3324–E3333. Bibcode:2017PNAS..114E3324S. doi:10.1073/pnas.1614557114. PMC 5402433. PMID 28351971.
- ^ "OMIM Entry - # 601472 - Charcot–Marie–Tooth disease, axonal, type 2D; CMT2D". Online Mendelian Inheritance in Man. Retrieved 2022-05-10.
- ^ "OMIM Entry- * 600287 - Glycl-tRNA Synthetase 1; GARS1". Online Mendelian Inheritance in Man. Retrieved 2022-05-10.
- ^ Wei N, Zhang Q, Yang XL (2019). "Neurodegenerative Charcot-Marie-Tooth disease as a case study to decipher novel functions of aminoacyl-tRNA synthetases". J Biol Chem. 294 (14): 5321–5339. doi:10.1074/jbc.REV118.002955. PMC 6462521. PMID 30643024.
- ^ a b He W, Bai G, Zhou H, Wei N, White NM, Lauer J, et al. (October 2015). "CMT2D neuropathy is linked to the neomorphic binding activity of glycyl-tRNA synthetase". Nature. 526 (7575): 710–714. Bibcode:2015Natur.526..710H. doi:10.1038/nature15510. PMC 4754353. PMID 26503042.
- ^ Mendonsa S, von Kuegelgen N, Bujanic L, Chekulaeva M (September 2021). "Charcot-Marie-Tooth mutation in glycyl-tRNA synthetase stalls ribosomes in a pre-accommodation state and activates integrated stress response". Nucleic Acids Research. 49 (17): 10007–10017. doi:10.1093/nar/gkab730. PMC 8464049. PMID 34403468.
- ^ a b Berger P, Young P, Suter U (March 2002). "Molecular cell biology of Charcot-Marie-Tooth disease". Neurogenetics. 4 (1): 1–15. doi:10.1007/s10048-002-0130-z. PMID 12030326.
- ^ Kleopa KA (December 2011). "The role of gap junctions in Charcot-Marie-Tooth disease". The Journal of Neuroscience. 31 (49): 17753–17760. doi:10.1523/JNEUROSCI.4824-11.2011. PMC 6634164. PMID 22159091.
- ^ Szigeti K, Lupski JR (June 2009). "Charcot-Marie-Tooth disease". European Journal of Human Genetics. 17 (6): 703–710. doi:10.1038/ejhg.2009.31. PMC 2947101. PMID 19277060.
- ^ Koutsis G, Breza M, Velonakis G, Tzartos J, Kasselimis D, Kartanou C, et al. (February 2019). "X linked Charcot-Marie-Tooth disease and multiple sclerosis: emerging evidence for an association". Journal of Neurology, Neurosurgery, and Psychiatry. 90 (2): 187–194. doi:10.1136/jnnp-2018-319014. PMID 30196252.
- ^ Amiott EA, Lott P, Soto J, Kang PB, McCaffery JM, DiMauro S, et al. (May 2008). "Mitochondrial Fusion and Function in Charcot-Marie-Tooth Type 2A Patient Fibroblasts with Mitofusin 2 Mutations". Experimental Neurology. 211 (1): 115–127. doi:10.1016/j.expneurol.2008.01.010. PMC 2409111. PMID 18316077.
- ^ Yiu EM, Burns J, Ryan MM, Ouvrier RA (September 2008). "Neurophysiologic abnormalities in children with Charcot-Marie-Tooth disease type 1A". Journal of the Peripheral Nervous System. 13 (3): 236–241. doi:10.1111/j.1529-8027.2008.00182.x. PMID 18844790.
- ^ "Diagnosing CMT". Charcot–Marie–Tooth Association. Retrieved 2020-05-30.
- ^ Wade N (2010-03-10). "Disease Cause Is Pinpointed With Genome". New York Times. Archived from the original on 2022-01-01.
- ^ Midroni G, Bilbao JM, Cohen SM (1995). Biopsy diagnosis of peripheral neuropathy. Boston: Butterworth-Heinemann. pp. 75–103. ISBN 978-0-7506-9552-7.
- ^ Dubowitz V, Sewry CA, Oldfors A, Lane R (2013). Muscle biopsy: a practical approach (Fourth ed.). Philadelphia: Saunders/Elsevier. ISBN 978-0-7020-4340-6.
- ^ "Medical Alert". Charcot-Marie-Tooth Association. Archived from the original on 2007-07-02. Retrieved 2007-08-21.
- ^ Anand N, Levine DB, Burke S, Bansal M (August 1997). "Neuropathic Spinal Arthropathy in Charcot-Marie-Tooth Disease. A Case Report". The Journal of Bone & Joint Surgery. 79 (8): 1235–9. doi:10.2106/00004623-199708000-00017. PMID 9278085.
- ^ Aguirre-Rodríguez FJ, Lucenilla MI, Alvarez-Cubero MJ, Mata C, Entrala-Bernal C, Fernandez-Rosado F (October 2015). "Novel FA2H mutation in a girl with familial spastic paraplegia". Journal of the Neurological Sciences. 357 (1–2): 332–334. doi:10.1016/j.jns.2015.08.1183. PMID 26344562.
- ^ Kobayashi T, Leung AK, Akazawa Y, Hutchins SW (March 2013). "The effect of varying the plantarflexion resistance of an ankle-foot orthosis on knee joint kinematics in patients with stroke". Gait & Posture. 37 (3): 457–459. doi:10.1016/j.gaitpost.2012.07.028. PMID 22921491.
- ^ Meyns P, Kerkum Y, Brehm M, Becher J, Buizer A, Harlaar J (May 2020). "Ankle foot orthoses in cerebral palsy: Effects of ankle stiffness on trunk kinematics, gait stability and energy cost of walking". European Journal of Paediatric Neurology. 26: 68–74. doi:10.1016/j.ejpn.2020.02.009. PMID 32147412.
- ^ "The effect of ankle foot orthosis stiffness on trunk movement and walking energy cost in cerebral palsy". Gait & Posture. 49: 2. September 2016. doi:10.1016/j.gaitpost.2016.07.070.
- ^ Kerkum YL, Buizer AI, van den Noort JC, Becher JG, Harlaar J, Brehm MA (2015-11-23). "The Effects of Varying Ankle Foot Orthosis Stiffness on Gait in Children with Spastic Cerebral Palsy Who Walk with Excessive Knee Flexion". PLOS ONE (in German). 10 (11): e0142878. Bibcode:2015PLoSO..1042878K. doi:10.1371/journal.pone.0142878. PMC 4658111. PMID 26600039.
- ^ Ploeger HE, Waterval NF, Nollet F, Bus SA, Brehm MA (2019). "Stiffness modification of two ankle-foot orthosis types to optimize gait in individuals with non-spastic calf muscle weakness - a proof-of-concept study". Journal of Foot and Ankle Research (in German). 12: 41. doi:10.1186/s13047-019-0348-8. PMC 6686412. PMID 31406508.
- ^ Pareyson D, Marchesi C (July 2009). "Diagnosis, natural history, and management of Charcot-Marie-Tooth disease". The Lancet. Neurology. 8 (7): 654–667. doi:10.1016/S1474-4422(09)70110-3. PMID 19539237.
- ^ Aboussouan LS, Lewis RA, Shy ME (2007-02-09). "Disorders of pulmonary function, sleep, and the upper airway in Charcot-Marie-Tooth disease". Lung. 185 (1): 1–7. doi:10.1007/s00408-006-0053-9. PMID 17294338.
External links
[edit] Media related to Charcot-Marie-Tooth disease at Wikimedia Commons
Media related to Charcot-Marie-Tooth disease at Wikimedia Commons



