Ulcerative colitis
| Ulcerative colitis | |
|---|---|
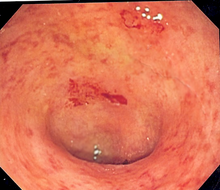 | |
| Endoscopic image of a colon affected by ulcerative colitis. The internal surface of the colon is blotchy and broken in places. Mild-moderate disease. | |
| Specialty | Gastroenterology |
| Symptoms | Abdominal pain, diarrhea mixed with blood, weight loss, fever, anemia,[1] dehydration, loss of appetite, fatigue, sores on the skin, urgency to defecate, inability to defecate despite urgency, rectal pain[2] |
| Complications | Megacolon, inflammation of the eye, joints, or liver, colon cancer[1][3] |
| Usual onset | 15–30 years or >60 years[1] |
| Duration | Long term[1] |
| Causes | Unknown[1] |
| Diagnostic method | Colonoscopy with tissue biopsies[1] |
| Differential diagnosis | Dysentery, Crohn's disease, ischemic colitis[4] |
| Treatment | Dietary changes, medication, surgery[1] |
| Medication | Sulfasalazine, mesalazine, steroids, immunosuppressants such as azathioprine, biological therapy[1] |
| Frequency | 2–299 per 100,000[5] |
| Deaths | 47,400 together with Crohn's (2015)[6] |
Ulcerative colitis (UC) is one of the two types of inflammatory bowel disease (IBD), with the other type being Crohn's disease.[1] It is a long-term condition that results in inflammation and ulcers of the colon and rectum.[1][7] The primary symptoms of active disease are abdominal pain and diarrhea mixed with blood (hematochezia).[1] Weight loss, fever, and anemia may also occur.[1] Often, symptoms come on slowly and can range from mild to severe.[1] Symptoms typically occur intermittently with periods of no symptoms between flares.[1] Complications may include abnormal dilation of the colon (megacolon), inflammation of the eye, joints, or liver, and colon cancer.[1][3]
The cause of UC is unknown.[1] Theories involve immune system dysfunction, genetics, changes in the normal gut bacteria, and environmental factors.[1][8] Rates tend to be higher in the developed world with some proposing this to be the result of less exposure to intestinal infections, or to a Western diet and lifestyle.[7][9] The removal of the appendix at an early age may be protective.[9] Diagnosis is typically by colonoscopy with tissue biopsies.[1]
Several medications are used to treat symptoms and bring about and maintain remission, including aminosalicylates such as mesalazine or sulfasalazine, steroids, immunosuppressants such as azathioprine, and biologic therapy.[1] Removal of the colon by surgery may be necessary if the disease is severe, does not respond to treatment, or if complications such as colon cancer develop.[1] Removal of the colon and rectum generally cures the condition.[1][9]
Signs and symptoms
[edit]| Crohn's disease | Ulcerative colitis | |
|---|---|---|
| Defecation | Often porridge-like,[10] sometimes steatorrhea |
Often mucus-like and with blood[10] |
| Tenesmus | Less common[10] | More common[10] |
| Fever | Common[10] | Indicates severe disease[10] |
| Fistulae | Common[11] | Seldom |
| Weight loss | Often | More seldom |
Gastrointestinal
[edit]People with ulcerative colitis usually present with diarrhea mixed with blood,[12] of gradual onset that persists for an extended period of time (weeks). It is estimated that 90% of people experience rectal bleeding (of varying severity), 90% experience watery or loose stools with increased stool frequency (diarrhea), and 75-90% of people experience bowel urgency.[13] Additional symptoms may include fecal incontinence, mucous rectal discharge, and nocturnal defecations.[12] With proctitis (inflammation of the rectum), people with UC may experience urgency or rectal tenesmus, which is the urgent desire to evacuate the bowels but with the passage of little stool.[12] Tenesmus may be misinterpreted as constipation, due to the urge to defecate despite small volume of stool passage. Bloody diarrhea and abdominal pain may be more prominent features in severe disease.[12] The severity of abdominal pain with UC varies from mild discomfort to very painful bowel movements and abdominal cramping.[14] High frequency of bowel movements, weight loss, nausea, fatigue, and fever are also common during disease flares. Chronic bleeding from the GI tract, chronic inflammation, and iron deficiency often leads to anemia, which can affect quality of life.[15]
The clinical presentation of ulcerative colitis depends on the extent of the disease process.[16] Up to 15% of individuals may have severe disease upon initial onset of symptoms.[12] A substantial proportion (up to 45%) of people with a history of UC without any ongoing symptoms (clinical remission) have objective evidence of ongoing inflammation.[17] Ulcerative colitis is associated with a generalized inflammatory process that can affect many parts of the body. Sometimes, these associated extra-intestinal symptoms are the initial signs of the disease.[18]
Extent of involvement
[edit]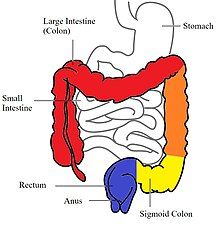
In contrast to Crohn's disease, which can affect areas of the gastrointestinal tract outside of the colon, ulcerative colitis is usually confined to the colon. Inflammation in ulcerative colitis is usually continuous, typically involving the rectum, with involvement extending proximally (to sigmoid colon, ascending colon, etc.).[19] In contrast, inflammation with Crohn's disease is often patchy, with so-called "skip lesions" (intermittent regions of inflamed bowel).[20]
The disease is classified by the extent of involvement, depending on how far the disease extends:[14] proctitis (rectal inflammation), left sided colitis (inflammation extending to descending colon), and extensive colitis (inflammation proximal to the descending colon).[19] Proctosigmoiditis describes inflammation of the rectum and sigmoid colon. Pancolitis describes involvement of the entire colon, extending from the rectum to the cecum. While usually associated with Crohn's disease, ileitis (inflammation of the ileum) also occurs in UC. About 17% of individuals with UC have ileitis.[21] Ileitis more commonly occurs in the setting of pancolitis (occurring in 20% of cases of pancolitis),[12] and tends to correlate with the activity of colitis. This so-called "backwash ileitis" can occur in 10–20% of people with pancolitis and is believed to be of little clinical significance.[22]
Severity of disease
[edit]In addition to the extent of involvement, UC is also characterized by severity of disease.[19] Severity of disease is defined by symptoms, objective markers of inflammation (endoscopic findings, blood tests), disease course, and the impact of the disease on day-to-day life.[19] Most patients are categorized through endoscopy and fecal calprotectin levels. Indicators of low risk for future complications in mild and moderate UC include the following parameters: exhibiting less than 6 stools daily and lack of fever/weight loss. Other indicators include lack of extraintestinal symptoms, low levels of the inflammatory markers C-reactive protein (CRP), and erythrocyte sedimentation rate (ESR), and fecal calprotectin, and later age of diagnosis (over 40 years).[23] Mild disease correlates with fewer than four stools daily; in addition, mild urgency and rectal bleeding may occur intermittently.[19] Mild disease lacks systemic signs of toxicity (e.g. fever, chills, weight changes) and exhibits normal levels of the serum inflammatory markers ESR and CRP.[23]
Moderate to severe disease correlates with more than six stools daily, frequent bloody stools and urgency.[19] Moderate abdominal pain, low-grade fever, 38 to 39 °C (100 to 102 °F), and anemia may develop.[19] ESR and CRP are usually elevated.[19]
The Mayo Score, which incorporates a combination of clinical symptoms (stool frequency and amount of rectal bleeding) with endoscopic findings and a physicians assessment of severity, is often used clinically to classify UC as mild, moderate or severe.[13]
Acute-Severe Ulcerative Colitis (ASUC) is a severe form which presents acutely and with severe symptoms. This fulminant type is associated with severe symptoms (usually diarrhea, rectal bleeding and abdominal pain) and is usually associated with systemic symptoms including fever.[13] It is associated with a high mortality rate as compared to milder forms of UC, with a 3-month and 12 month mortality rate of 0.84% and 1% respectively.[13] People with fulminant UC may have inflammation extending beyond just the mucosal layer, causing impaired colonic motility and leading to toxic megacolon. Toxic megacolon represents a medical emergency, one often treated surgically. If the serous membrane is involved, a colonic perforation may ensue, which has a 50% mortality rate in people with UC.[24] Other complications include hemorrhage, venous thromboembolism, and secondary infections of the colon including C. difficile or cytomegalovirus colitis.[13]
Ulcerative colitis may improve and enter remission.[19]
Extraintestinal manifestations and complications
[edit]| Crohn's disease |
Ulcerative colitis | ||
|---|---|---|---|
| Nutrient deficiency | Higher risk | ||
| Colon cancer risk | Slight | Considerable | |
| Prevalence of extraintestinal complications[25][26][27] | |||
| Iritis/uveitis | Females | 2.2% | 3.2% |
| Males | 1.3% | 0.9% | |
| Primary sclerosing cholangitis |
Females | 0.3% | 1% |
| Males | 0.4% | 3% | |
| Ankylosing spondylitis |
Females | 0.7% | 0.8% |
| Males | 2.7% | 1.5% | |
| Pyoderma gangrenosum |
Females | 1.2% | 0.8% |
| Males | 1.3% | 0.7% | |
| Erythema nodosum | Females | 1.9% | 2% |
| Males | 0.6% | 0.7% | |

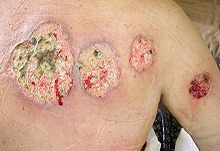
UC is characterized by immune dysregulation and systemic inflammation, which may result in symptoms and complications outside the colon. Commonly affected organs include: eyes, joints, skin, and liver.[28] The frequency of such extraintestinal manifestations has been reported as between 6 and 47%.[29][30]
UC may affect the mouth. About 8% of individuals with UC develop oral manifestations.[31] The two most common oral manifestations are aphthous stomatitis and angular cheilitis.[31] Aphthous stomatitis is characterized by ulcers in the mouth, which are benign, noncontagious and often recurrent. Angular chelitis is characterized by redness at the corners of the mouth, which may include painful sores or breaks in the skin.[31] Very rarely, benign pustules may occur in the mouth (pyostomatitis vegetans).[31]
UC may affect the eyes manifesting in scleritis, iritis, and conjunctivitis. Patients may be asymptomatic or experience redness, burning, or itching in eyes. Inflammation may occur in the interior portion of the eye, leading to uveitis and iritis.[32] Uveitis can cause blurred vision and eye pain, especially when exposed to light (photophobia). Untreated, uveitis can lead to permanent vision loss.[32] Inflammation may also involve the white part of the eye (sclera) or the overlying connective tissue (episclera), causing conditions called scleritis and episcleritis.[33] Ulcerative colitis is most commonly associated with uveitis and episcleritis.[34]
UC may cause several joint manifestations, including a type of rheumatologic disease known as seronegative arthritis, which may affect few large joints (oligoarthritis), the vertebra (ankylosing spondylitis) or several small joints of the hands and feet (peripheral arthritis).[28] Often the insertion site where muscle attaches to bone (entheses) becomes inflamed (enthesitis). Inflammation may affect the sacroiliac joint (sacroiliitis).[18] It is estimated that around 50% of IBD patients suffer from migratory arthritis. Synovitis, or inflammation of the synovial fluid surrounding a joint, can occur for months and recur in later times but usually does not erode the joint. The symptoms of arthritis include joint pain, swelling, and effusion, and often leads to significant morbidity.[18] Ankylosing spondylitis and sacroilitis usually occur independent of bowel disease activity in UC.[13]
Ulcerative colitis may affect the skin. The most common type of skin manifestation, erythema nodosum, presents in up to 3% of UC patients. It develops as raised, tender red nodules usually appearing on the outer areas of the arms or legs, especially in the anterior tibial area (shins).[34] The nodules have diameters that measure approximately 1–5 cm. Erythema nodosum is due to inflammation of the underlying subcutaneous tissue (panniculitis), and biopsy will display focal panniculitis (although is often unnecessary in diagnosis). In contrast to joint-related manifestations, erythema nodosum often occurs alongside intestinal disease. Thus, treatment of UC can often lead to resolution of skin nodules.[35]
Another skin condition associated with UC is pyoderma gangrenosum, which presents as deep skin ulcerations. Pyoderma gangrenosum is seen in about 1% of patients with UC and its formation is usually independent of bowel inflammation.[13] Pyoderma gangrenosum is characterized by painful lesions or nodules that become ulcers which progressively grow. The ulcers are often filled with sterile pus-like material. In some cases, pyoderma gangrenosum may require injection with corticosteroids.[28] Treatment may also involve inhibitors of tumor necrosis factor (TNF), a cytokine that promotes cell survival.[35]
Other associations determined between the skin and ulcerative colitis include a skin condition known as hidradenitis suppurativa (HS). This condition represents a chronic process in which follicles become occluded leading to recurring inflammation of nodules and abscesses and even fistulas tunnels in the skin that drain fluid.[36]
Ulcerative colitis may affect the circulatory and endocrine system. UC increases the risk of blood clots in both arteries and veins;[37][38][39] painful swelling of the lower legs can be a sign of deep venous thrombosis, while difficulty breathing may be a result of pulmonary embolism (blood clots in the lungs). The risk of blood clots is about threefold higher in individuals with IBD.[38] The risk of venous thromboembolism is high in ulcerative colitis due to hypercoagulability from inflammation, especially with active or extensive disease.[37] Additional risk factors may include surgery, hospitalization, pregnancy, the use of corticosteroids and tofacitinib, a JAK inhibitor.[37]
Osteoporosis may occur related to systemic inflammation or prolonged steroid use in the treatment of UC, which increases the risk of bone fractures.[18] Clubbing, a deformity of the ends of the fingers, may occur.[18] Amyloidosis may occur, especially with severe and poorly controlled disease, which usually presents with protein in the urine (proteinuria) and nephritic syndrome.[18]
Primary sclerosing cholangitis
[edit]Ulcerative colitis (UC) has a significant association with primary sclerosing cholangitis (PSC), a progressive inflammatory disorder of small and large bile ducts. Up to 70-90% of people with primary sclerosing cholangitis have ulcerative colitis.[34] As many as 5% of people with UC may progress to develop primary sclerosing cholangitis.[28][40] PSC is more common in men, and often begins between 30 and 40 years of age.[28] It can present asymptomatically or exhibit symptoms of itchiness (pruritis) and fatigue. Other symptoms include systemic signs such as fever and night sweats. Such symptoms are often associated with a bacterial episodic version of PSC. Upon physical exam, one may discern enlarged liver contours (hepatomegaly) or enlarged spleen (splenomegaly) as well as areas of excoriation. Yellow coloring of the skin, or jaundice, may also be present due to excess of bile byproduct buildup (bilirubin) from the biliary tract.
In diagnosis, lab results often reveal a pattern indicative of biliary disease (cholestatic pattern). This is often displayed by markedly elevated alkaline phosphatase levels and milder or no elevation in liver enzyme levels. Results of endoscopic retrograde cholangiography (ERC) may show bile ducts with thicker walls, areas of dilation or narrowing. However, some patients with UC and PSC have inflammation that has significantly affected only ramified intrahepatic bile ducts of smaller diameter, also known as "small ducts", which are not visualized by ERC.[41]: 604, 609
In some cases, primary sclerosing cholangitis occurs several years before the bowel symptoms of ulcerative colitis develop.[34] PSC does not parallel the onset, extent, duration, or activity of the colonic inflammation in ulcerative colitis.[34] In addition, colectomy does not have an impact on the course of primary sclerosing cholangitis in individuals with UC.[34] PSC is associated with an increased risk of colorectal cancer and cholangiocarcinoma (bile duct cancer).[34][28] PSC is a progressive condition, and may result in cirrhosis of the liver.[28] No specific therapy has been proven to affect the long-term course of PSC.[28]
Causes
[edit]| Crohn's disease | Ulcerative colitis | |
|---|---|---|
| Smoking | Higher risk for smokers | Lower risk for smokers[19] |
| Age | Usual onset between 15 and 30 years[42] |
Peak incidence between 15 and 25 years |
Ulcerative colitis is an autoimmune disease characterized by T-cells infiltrating the colon.[43] No direct causes for UC are known, but factors such as genetics, environment, and an overactive immune system play a role.[1] UC is associated with comorbidities that produce symptoms in many areas of the body outside the digestive system.
Genetic factors
[edit]A genetic component to the cause of UC can be hypothesized based on aggregation of UC in families, variation of prevalence between different ethnicities, genetic markers and linkages.[44] In addition, the identical twin concordance rate is 10%, whereas the dizygotic twin concordance rate is only 3%.[44][45] Between 8 and 14% of people with ulcerative colitis have a family history of inflammatory bowel disease.[12] In addition, people with a first degree relative with UC have a four-fold increase in their risk of developing the disease.[12]
Twelve regions of the genome may be linked to UC, including, in the order of their discovery, chromosomes 16, 12, 6, 14, 5, 19, 1, and 3,[46] but none of these loci has been consistently shown to be at fault, suggesting that the disorder is influenced by multiple genes. For example, chromosome band 1p36 is one such region thought to be linked to inflammatory bowel disease.[47] Some of the putative regions encode transporter proteins such as OCTN1 and OCTN2. Other potential regions involve cell scaffolding proteins such as the MAGUK family. Human leukocyte antigen associations may even be at work. In fact, this linkage on chromosome 6 may be the most convincing and consistent of the genetic candidates.[46]
Multiple autoimmune disorders are associated with ulcerative colitis, including celiac disease,[48] psoriasis,[49] lupus erythematosus,[50] rheumatoid arthritis,[51] episcleritis, and scleritis.[32] Ulcerative colitis is also associated with acute intermittent porphyria.[52]
Environmental factors
[edit]Many hypotheses have been raised for environmental factors contributing to the pathogenesis of ulcerative colitis, including diet, breastfeeding and medications. Breastfeeding may have a protective effect in the development of ulcerative colitis.[53][54] One study of isotretinoin found a small increase in the rate of UC.[55]
As the colon is exposed to many dietary substances which may encourage inflammation, dietary factors have been hypothesized to play a role in the pathogenesis of both ulcerative colitis and Crohn's disease. However, research does not show a link between diet and the development of ulcerative colitis. Few studies have investigated such an association; one study showed no association of refined sugar on the number of people affected of ulcerative colitis.[56] High intake of unsaturated fat and vitamin B6 may enhance the risk of developing ulcerative colitis.[57] Other identified dietary factors that may influence the development and/or relapse of the disease include meat protein and alcoholic beverages.[58][59] Specifically, sulfur has been investigated as being involved in the cause of ulcerative colitis, but this is controversial.[60] Sulfur restricted diets have been investigated in people with UC and animal models of the disease. The theory of sulfur as an etiological factor is related to the gut microbiota and mucosal sulfide detoxification in addition to the diet.[61][62][63]
As a result of a class-action lawsuit and community settlement with DuPont, three epidemiologists conducted studies on the population surrounding a chemical plant that was exposed to PFOA at levels greater than in the general population. The studies concluded that there was an association between PFOA exposure and six health outcomes, one of which being ulcerative colitis.[64]
Alternative theories
[edit]Levels of sulfate-reducing bacteria tend to be higher in persons with ulcerative colitis, which could indicate higher levels of hydrogen sulfide in the intestine. An alternative theory suggests that the symptoms of the disease may be caused by toxic effects of the hydrogen sulfide on the cells lining the intestine.[65]
Infection by Mycobacterium avium, subspecies paratuberculosis, has been proposed as the ultimate cause of both ulcerative colitis and Crohn's disease.[66]
Pathophysiology
[edit]| Crohn's disease | Ulcerative colitis | |
|---|---|---|
| Cytokine response | Associated with Th17[67] | Vaguely associated with Th2 |
An increased amount of colonic sulfate-reducing bacteria has been observed in some people with ulcerative colitis, resulting in higher concentrations of the toxic gas hydrogen sulfide. Human colonic mucosa is maintained by the colonic epithelial barrier and immune cells in the lamina propria (see intestinal mucosal barrier). The short-chain fatty acid n-butyrate gets oxidized through the beta oxidation pathway into carbon dioxide and ketone bodies. It has been shown that n-butyrate helps supply nutrients to this epithelial barrier. Studies have proposed that hydrogen sulfide plays a role in impairing this beta-oxidation pathway by interrupting the short chain acetyl-CoA dehydrogenase, an enzyme within the pathway. Furthermore, it has been suggested that the protective effect of smoking in ulcerative colitis is due to the hydrogen cyanide from cigarette smoke reacting with hydrogen sulfide to produce the non-toxic isothiocyanate, thereby inhibiting sulfides from interrupting the pathway.[68] An unrelated study suggested that the sulfur contained in red meats and alcohol may lead to an increased risk of relapse for people in remission.[65]
Other proposed mechanisms driving the pathophysiology of ulcerative colitis involve an abnormal immune response to the normal gut microbiota. This involves abnormal activity of antigen presenting cells (APCs) including dendritic cells and macrophages. Normally, dendritic cells and macrophages patrol the intestinal epithelium and phagocytose (engulf and destroy) pathogenic microorganisms and present parts of the microorganism as antigens to T-cells to stimulate differentiation and activation of the T-cells.[13] However, in ulcerative colitis, aberrant activity of dendritic cells and macrophages results in them phagocytosing bacteria of the normal gut microbiome. After ingesting the microbiome bacterium, the APCs release the cytokine TNFα which stimulates inflammatory signaling and recruits inflammatory cells to the intestines, leading to the inflammation that is characteristic of ulcerative colitis.[13] The TNF inhibitors, including infliximab, adalimumab and golimumab, are used to inhibit this step during the treatment of ulcerative colitis.[13] After phagocytosing the microbe, the APCs then enter the mesenteric lymph nodes where they present antigens to naive T-cells while also releasing the pro-inflammatory cytokines IL-12 and IL-23 which lead to T cell differentiation into Th1 and Th17 T-cells.[13] IL-12 and IL-23 signaling is blocked by the biologic ustekinumab and IL-23 is blocked by guselkumab, mirikizumab and risankizumab, medications that are used in the treatment of ulcerative colitis.[13] From the mesenteric lymph node, the T-cells then enter the intestinal lymphatic venule which provides transport to the intestinal epithelium where they mediate further inflammation characteristic of ulcerative colitis.[13] The T-cells exit the lymphatic venule via the adhesion protein mucosal vascular addressin cell adhesion molecule 1 MAdCAM-1, the ulcerative colitis biologic treatment vedolizumab inhibits T-cell migration out of the lymphatic venules by blocking binding to MAdCAM-1.[13] While the medications ozanimod and etrasimod inhibit the sphingosine-1-phosphate receptor to prevent T-cell migration into the efferent lymphatic venules.[13] Once the mature Th1 and Th17 T-cells exit the efferent lymphatic venule, they travel to the intestinal mucosa and cause further inflammation. T-cell mediated inflammation is thought to be driven by the JAK-STAT intracellular T-cell signaling pathway, leading to the transcription, translation and release of inflammatory cytokines. This T-cell JAK-STAT signaling is inhibited by the medications tofacitinib, filgotinib and upadacitinib which are used in the treatment of ulcerative colitis.[13]
Diagnosis
[edit]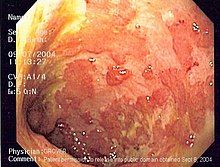

The initial diagnostic workup for ulcerative colitis consists of a complete history and physical examination, assessment of signs and symptoms, laboratory tests and endoscopy.[69] Severe UC can exhibit high erythrocyte sedimentation rate (ESR), decreased albumin (a protein produced by the liver), and various changes in electrolytes. As discussed previously, UC patients often also display elevated alkaline phosphatase. Inflammation in the intestine may also cause higher levels of fecal calprotectin or lactoferrin.[70]
Specific testing may include the following:[19][71]
- A complete blood count is done to check for anemia; thrombocytosis, a high platelet count, is occasionally seen
- Electrolyte studies and kidney function tests are done, as chronic diarrhea may be associated with hypokalemia, hypomagnesemia and kidney injury.
- Liver function tests are performed to screen for bile duct involvement: primary sclerosing cholangitis.
- Imaging such as x-ray or CT scan to evaluate for possible perforation or toxic megacolon
- Stool culture and Clostridioides difficile stool assay to rule out infectious colitis[69]
- Inflammatory markers, such as erythrocyte sedimentation rate or C-reactive protein
- Lower endoscopy to evaluate the rectum and distal large intestine (sigmoidoscopy) or entire colon and end of the small intestine (colonoscopy) for ulcers and inflammation
Although ulcerative colitis is a disease of unknown causation, inquiry should be made as to unusual factors believed to trigger the disease.[19]
The simple clinical colitis activity index was created in 1998 and is used to assess the severity of symptoms.[72]
Endoscopic
[edit]
The best test for diagnosis of ulcerative colitis remains endoscopy, which is examination of the internal surface of the bowel using a flexible camera. Initially, a flexible sigmoidoscopy may be completed to establish the diagnosis.[73] The physician may elect to limit the extent of the initial exam if severe colitis is encountered to minimize the risk of perforation of the colon. However, a complete colonoscopy with entry into the terminal ileum should be performed to rule out Crohn's disease, and assess extent and severity of disease.[73] Endoscopic findings in ulcerative colitis include: erythema (redness of the mucosa), friability of the mucosa, superficial ulceration, and loss of the vascular appearance of the colon. When present, ulcerations may be confluent. Pseudopolyps may be observed.[74]
Ulcerative colitis is usually continuous from the rectum, with the rectum almost universally being involved. Perianal disease is rare. The degree of involvement endoscopically ranges from proctitis (rectal inflammation) to left sided colitis (extending to descending colon), to extensive colitis (extending proximal to descending colon).[14]
Histologic
[edit]

Biopsies of the mucosa are taken during endoscopy to confirm the diagnosis of UC and differentiate it from Crohn's disease, which is managed differently clinically. Histologic findings in ulcerative colitis includes: distortion of crypt architecture, crypt abscesses, and inflammatory cells in the mucosa (lymphocytes, plasma cells, and granulocytes).[28] Unlike the transmural inflammation seen in Crohn's disease, the inflammation of ulcerative colitis is limited to the mucosa.[28]
Laboratory tests
[edit]Blood and stool tests serve primarily to assess disease severity, level of inflammation and rule out causes of infectious colitis. All individuals with suspected ulcerative colitis should have stool testing to rule out infection.[12]
A complete blood count may demonstrate anemia, leukocytosis, or thrombocytosis.[12] Anemia may be caused by inflammation or bleeding. Chronic blood loss may lead to iron deficiency as a cause for anemia, particularly microcytic anemia (small red blood cells), which can be evaluated with a serum ferritin, iron, total iron-binding capacity and transferrin saturation. Anemia may be due to a complication of treatment from azathioprine, which can cause low blood counts,[75] or sulfasalazine, which can result in folate deficiency. Thiopurine metabolites (from azathioprine) and a folate level can help.[76]
UC may cause high levels of inflammation throughout the body, which may be quantified with serum inflammatory markers, such as CRP and ESR. However, elevated inflammatory markers are not specific for UC and elevations are commonly seen in other conditions, including infection. In addition, inflammatory markers are not uniformly elevated in people with ulcerative colitis. Twenty five percent of individuals with confirmed inflammation on endoscopic evaluation have a normal CRP level.[19] Serum albumin may also be low related to inflammation, in addition to loss of protein in the GI tract associated with bleeding and colitis. Low serum levels of vitamin D are associated with UC, although the significance of this finding is unclear.[77]
Specific antibody markers may be elevated in ulcerative colitis. Specifically, perinuclear antineutrophil cytoplasmic antibodies (pANCA) are found in 70 percent of cases of UC.[19] Antibodies against Saccharomyces cerevisiae may be present, but are more often positive in Crohn's disease compared with ulcerative colitis. However, due to poor accuracy of these serolologic tests, they are not helpful in the diagnostic evaluation of possible inflammatory bowel disease.[19][28]
Several stool tests may help quantify the extent of inflammation present in the colon and rectum. Fecal calprotectin is elevated in inflammatory conditions affecting the colon, and is useful in distinguishing irritable bowel syndrome (noninflammatory) from a flare in inflammatory bowel disease.[19] Fecal calprotectin is 88% sensitive and 79% specific for the diagnosis of ulcerative colitis.[19] If the fecal calprotectin is low, the likelihood of inflammatory bowel disease are less than 1 percent.[12] Lactoferrin is an additional nonspecific marker of intestinal inflammation.[78]
Imaging
[edit]Overall, imaging tests, such as x-ray or CT scan, may be helpful in assessing for complications of ulcerative colitis, such as perforation or toxic megacolon. Bowel ultrasound (US) is a cost-effective, well-tolerated, non-invasive and readily available tool for the management of patients with inflammatory bowel disease (IBD), including UC, in clinical practice.[79] Some studies demonstrated that bowel ultrasound is an accurate tool for assessing disease activity in people with ulcerative colitis.[80][81] Imaging is otherwise of limited use in diagnosing ulcerative colitis.[12][28] Magnetic resonance imaging (MRI) is necessary to diagnose underlying PSC.[28]
Abdominal xray is often the test of choice and may display nonspecific findings in cases of mild or moderate ulcerative colitis. In circumstances of severe UC, radiographic findings may include thickening of the mucosa, often termed "thumbprinting", which indicates swelling due to fluid displacement (edema). Other findings may include colonic dilation and stool buildup evidencing constipation.[70]
Similar to xray, in mild ulcerative colitis, double contrast barium enema often shows nonspecific findings. Conversely, barium enema may display small buildups of barium in microulcerations. Severe UC can be characterized by various polyps, colonic shortening, loss of haustrae (the small bulging pouches in the colon), and narrowing of the colon. It is important to note that barium enema should not be conducted in patients exhibiting very severe symptoms as this may slow or stop stool passage through the colon causing ileus and toxic megacolon.[70]
Other methods of imaging include computed tomography (CT) and magnetic resonance imaging (MRI). Both may depict colonic wall thickening but have decreased ability to find early signs of wall changes when compared to barium enema. In cases of severe ulcerative colitis, however, they often exhibit equivalent ability to detect colonic changes.[70]
Doppler ultrasound is the last means of imaging that may be used. Similar to the imaging methods mentioned earlier, this may show some thickened bowel wall layers. In severe cases, this may show thickening in all bowel wall layers (transmural thickness).[70]
Differential diagnosis
[edit]Several conditions may present in a similar manner as ulcerative colitis and should be excluded. Such conditions include: Crohn's disease, infectious colitis, nonsteroidal anti-inflammatory drug enteropathy, and irritable bowel syndrome. Alternative causes of colitis should be considered, such as ischemic colitis (inadequate blood flow to the colon), radiation colitis (if prior exposure to radiation therapy), or chemical colitis. Pseudomembranous colitis may occur due to Clostridioides difficile infection following administration of antibiotics. Entamoeba histolytica is a protozoan parasite that causes intestinal inflammation. A few cases have been misdiagnosed as UC with poor outcomes occurring due to the use of corticosteroids.[82]
The most common disease that mimics the symptoms of ulcerative colitis is Crohn's disease, as both are inflammatory bowel diseases that can affect the colon with similar symptoms. It is important to differentiate these diseases since their courses and treatments may differ. In some cases, however, it may not be possible to tell the difference, in which case the disease is classified as indeterminate colitis.[83] Crohn's disease can be distinguished from ulcerative colitis in several ways. Characteristics that indicate Crohn's include evidence of disease around the anus (perianal disease). This includes anal fissures and abscesses as well as fistulas, which are abnormal connections between various bodily structures.[84]
Infectious colitis is another condition that may present in similar manner to ulcerative colitis. Endoscopic findings are also oftentimes similar. One can discern whether a patient has infectious colitis by employing tissue cultures and stool studies. Biopsy of the colon is another beneficial test but is more invasive.
Other forms of colitis that may present similarly include radiation and diversion colitis. Radiation colitis occurs after irradiation and often affects the rectum or sigmoid colon, similar to ulcerative colitis. Upon histology radiation colitis may indicate eosinophilic infiltrates, abnormal epithelial cells, or fibrosis. Diversion colitis, on the other hand, occurs after portions of bowel loops have been removed. Histology in this condition often shows increased growth of lymphoid tissue.
In patients who have undergone transplantation, graft versus host disease may also be a differential diagnosis. This response to transplantation often causes prolonged diarrhea if the colon is affected. Typical symptoms also include rash. Involvement of the upper gastrointestinal tract may lead to difficulty swallowing or ulceration. Upon histology, graft versus host disease may present with crypt cell necrosis and breakdown products within the crypts themselves.[85]
| Crohn's disease | Ulcerative colitis | |
|---|---|---|
| Terminal ileum involvement | Commonly | Seldom |
| Colon involvement | Usually | Always |
| Rectum involvement | Seldom | Usually (95%)[19] |
| Involvement around the anus |
Common[86] | Seldom |
| Bile duct involvement | No increase in rate of primary sclerosing cholangitis | Higher rate[87] |
| Distribution of disease | Patchy areas of inflammation (skip lesions) | Continuous area of inflammation[19] |
| Endoscopy | Deep geographic and serpiginous (snake-like) ulcers | Continuous ulcer |
| Depth of inflammation | May be transmural, deep into tissues[86][46] | Shallow, mucosal |
| Stenosis | Common | Seldom |
| Granulomas on biopsy | May have non-necrotizing non-peri-intestinal crypt granulomas[86][88][89] | Non-peri-intestinal crypt granulomas not seen[90] |
Management
[edit]Standard treatment for ulcerative colitis depends on the extent of involvement and disease severity. The goal is to induce remission initially with medications, followed by the administration of maintenance medications to prevent a relapse. The concept of induction of remission and maintenance of remission is very important. The medications used to induce and maintain a remission somewhat overlap, but the treatments are different. Physicians first direct treatment to inducing remission, which involves relief of symptoms and mucosal healing of the colon's lining, and then longer-term treatment to maintain remission and prevent complications.[91]
For acute stages of the disease, a low fiber diet may be recommended.[92][93][94]
Medication
[edit]The first-line maintenance medication for ulcerative colitis in remission is mesalazine (also known as mesalamine or 5-ASA).[95][96] For patients with active disease limited to the left colon (descending colon) or proctitis, mesalazine is also the first-line agent, and a combination of suppositories and oral mesalazine may be tried. Adding corticosteroids such as prednisone is also common in active disease, especially if remission is not achieved through mesalazine monotherapy,[95][96] but they are not used in long-term treatment as their risks then outweigh their benefits. Immunosuppressive medications such as azathioprine and biological agents such as infliximab, adalimumab, ustekinumab, vedolizumab, or risankizumab are given in severe disease or if a patient cannot achieve remission with mesalazine and corticosteroids.[97][98] As an alternative to mesalazine, one of its prodrugs such as sulfasalazine may be chosen for treatment of active disease or maintenance therapy,[99] but the prodrugs have greater potential for serious side effects and have not been demonstrated to be superior to mesalazine in large trials.[100]
A formulation of budesonide was approved by the U.S. Food and Drug Administration (FDA) for treatment of active ulcerative colitis in January 2013.[101][102] In 2018, tofacitinib was approved for treatment of moderately to severely active ulcerative colitis in the United States, the first oral medication indicated for long term use in this condition.[103] The evidence on methotrexate does not show a benefit in producing remission in people with ulcerative colitis.[104] Cyclosporine is effective for severe UC[97] and tacrolimus has also shown benefits.[105][106][107][108] Etrasimod was approved for medical use in the United States in October 2023.[109]
Aminosalicylates
[edit]Sulfasalazine has been a major agent in the therapy of mild to moderate ulcerative colitis for over 50 years. In 1977, it was shown that 5-aminosalicylic acid (5-ASA, mesalazine/mesalamine) was the therapeutically active component in sulfasalazine.[110] Many 5-ASA drugs have been developed with the aim of delivering the active compound to the large intestine to maintain therapeutic efficacy but with reduction of the side effects associated with the sulfapyridine moiety in sulfasalazine. Oral 5-ASA drugs are particularly effective in inducing and in maintaining remission in mild to moderate ulcerative colitis.[111][112] Rectal suppository, foam or liquid enema formulations of 5-ASA are used for colitis affecting the rectum, sigmoid or descending colon, and have been shown to be effective especially when combined with oral treatment.[113]
Biologics
[edit]Biologic treatments such as the TNF inhibitors infliximab, adalimumab, and golimumab are commonly used to treat people with UC who are no longer responding to corticosteroids. Tofacitinib and vedolizumab can also produce good clinical remission and response rates in UC.[8] Biologics may be used early in treatment (step down approach), or after other treatments have failed to induce remission (step up approach); the strategy should be individualized.[114]
Unlike aminosalicylates, biologics can cause serious side effects such as an increased risk of developing extra-intestinal cancers,[115] heart failure; and weakening of the immune system, resulting in a decreased ability of the immune system to clear infections and reactivation of latent infections such as tuberculosis. For this reason, people on these treatments are closely monitored and are often tested for hepatitis and tuberculosis annually.[116][117]
Etrasimod, a once-daily oral sphingosine 1-phosphate (S1P) receptor modulator that selectively activates S1P receptor subtypes 1, 4, and 5 with no detectable activity on S1P 2 or 3, is in development for treatment of immune-mediated diseases, including ulcerative colitis, and was shown in 2 randomized trials to be effective and well tolerated as induction and maintenance therapy in patients with moderately to severely active ulcerative colitis.[118]
Nicotine
[edit]Unlike Crohn's disease, ulcerative colitis has a lesser chance of affecting smokers than non-smokers.[119][120] In select individuals with a history of previous tobacco use, resuming low dose smoking may improve signs and symptoms of active ulcerative colitis,[121] but it is not recommended due to the overwhelmingly negative health effects of tobacco.[122] Studies using a transdermal nicotine patch have shown clinical and histological improvement.[123] In one double-blind, placebo-controlled study conducted in the United Kingdom, 48.6% of people with UC who used the nicotine patch, in conjunction with their standard treatment, showed complete resolution of symptoms. Another randomized, double-blind, placebo-controlled, single-center clinical trial conducted in the United States showed that 39% of people who used the patch showed significant improvement, versus 9% of those given a placebo.[124] However, nicotine therapy is generally not recommended due to side effects and inconsistent results.[125][126][127]
Iron supplementation
[edit]The gradual loss of blood from the gastrointestinal tract, as well as chronic inflammation, often leads to anemia, and professional guidelines suggest routinely monitoring for anemia with blood tests repeated every three months in active disease and annually in quiescent disease.[128] Adequate disease control usually improves anemia of chronic disease, but iron deficiency anemia should be treated with iron supplements. The form in which treatment is administered depends both on the severity of the anemia and on the guidelines that are followed. Some advise that parenteral iron be used first because people respond to it more quickly, it is associated with fewer gastrointestinal side effects, and it is not associated with compliance issues.[129] Others require oral iron to be used first, as people eventually respond and many will tolerate the side effects.[128][130]
Anticholinergics
[edit]Anticholinergic drugs, more specifically muscarinic antagonists, are sometimes used to treat abdominal cramps in connection with ulcerative colitis through their calming effect on colonic peristalsis (reducing both amplitude and frequency) and intestinal tone.[131][132] Some medical authorities suggest over-the-counter anticholinergic drugs as potential helpful treatments for abdominal cramping in mild ulcerative colitis.[133] However, their use is contraindicated especially in moderate to severe disease states because of the potential for anticholinergic treatment to induce toxic megacolon in patients with colonic inflammation.[134] Toxic megacolon is a state in which the colon is abnormally distended, and may in severe or untreated cases lead to colonic perforation, sepsis, and death.[135]
Immunosuppressant therapies, infection risks and vaccinations
[edit]Many patients affected by ulcerative colitis need immunosuppressant therapies, which may be associated with a higher risk of contracting opportunistic infectious diseases.[136] Many of these potentially harmful diseases, such as Hepatitis B, Influenza, chickenpox, herpes zoster virus, pneumococcal pneumonia, or human papilloma virus, can be prevented by vaccines. Each drug used in the treatment of IBD should be classified according to the degree of immunosuppression induced in the patient. Several guidelines suggest investigating patients' vaccination status before starting any treatment and performing vaccinations against vaccine preventable diseases when required.[137][138] Compared to the rest of the population, patients affected by IBD are known to be at higher risk of contracting some vaccine-preventable diseases.[139] Patients treated with Janus kinase inhibitor showed higher risk of Shingles.[140] Nevertheless, despite the increased risk of infections, vaccination rates in IBD patients are known to be suboptimal and may also be lower than vaccination rates in the general population.[141][142]
Surgery
[edit]| Crohn's disease | Ulcerative colitis | |
|---|---|---|
| Mesalazine | Less useful[143] | More useful[143] |
| Antibiotics | Effective in long-term[144] | Generally not useful[145] |
| Surgery | Often returns following removal of affected part |
Usually cured by removal of colon |
Unlike in Crohn's disease, the gastrointestinal aspects of ulcerative colitis can generally be cured by surgical removal of the large intestine, though extraintestinal symptoms may persist. This procedure is necessary in the event of: exsanguinating hemorrhage, frank perforation, or documented or strongly suspected carcinoma. Surgery is also indicated for people with severe colitis or toxic megacolon. People with symptoms that are disabling and do not respond to drugs may wish to consider whether surgery would improve the quality of life.[14]
The removal of the entire large intestine, known as a proctocolectomy, results in a permanent ileostomy – where a stoma is created by pulling the terminal ileum through the abdomen. Intestinal contents are emptied into a removable ostomy bag which is secured around the stoma using adhesive.[146]
Another surgical option for ulcerative colitis that is affecting most of the large bowel is called the ileal pouch-anal anastomosis (IPAA). This is a two- or three-step procedure. In a three-step procedure, the first surgery is a sub-total colectomy, in which the large bowel is removed, but the rectum remains in situ, and a temporary ileostomy is made. The second step is a proctectomy and formation of the ileal pouch (commonly known as a "j-pouch"). This involves removing the large majority of the remaining rectal stump and creating a new "rectum" by fashioning the end of the small intestine into a pouch and attaching it to the anus. After this procedure, a new type of ileostomy is created (known as a loop ileostomy) to allow the anastomoses to heal. The final surgery is a take-down procedure where the ileostomy is reversed and there is no longer the need for an ostomy bag. When done in two steps, a proctocolectomy – removing both the colon and rectum – is performed alongside the pouch formation and loop ileostomy. The final step is the same take-down surgery as in the three-step procedure. Time taken between each step can vary, but typically a six- to twelve-month interval is recommended between the first two steps, and a minimum of two to three months is required between the formation of the pouch and the ileostomy take-down.[14]
While the ileal pouch procedure removes the need for an ostomy bag, it does not restore normal bowel function. In the months following the final operation, patients typically experience 8–15 bowel movements a day. Over time this number decreases, with many patients reporting 4–6 bowel movements after one year post-op. While many patients have success with this procedure, there are a number of known complications. Pouchitis, inflammation of the ileal pouch resulting in symptoms similar to ulcerative colitis, is relatively common. Pouchitis can be acute, remitting, or chronic however treatment using antibiotics, steroids, or biologics can be highly effective. Other complications include fistulas, abscesses, and pouch failure. Depending on the severity of the condition, pouch revision surgery may need to be performed. In some cased the pouch may need to be de-functioned or removed and an ileostomy recreated.[147][148]
The risk of cancer arising from an ileal pouch anal anastomosis is low.[149] However, annual surveillance with pouchoscopy may be considered in individuals with risk factors for dysplasia, such as a history of dysplasia or colorectal cancer, a history of PSC, refractory pouchitis, and severely inflamed atrophic pouch mucosa.[149]
Bacterial recolonization
[edit]In a number of randomized clinical trials, probiotics have demonstrated the potential to be helpful in the treatment of ulcerative colitis. Specific types of probiotics such as Escherichia coli Nissle have been shown to induce remission in some people for up to a year.[150]
A Cochrane review of controlled trials using various probiotics found low-certainty evidence that probiotic supplements may increase the probability of clinical remission.[151] People receiving probiotics were 73% more likely to experience disease remission and over 2x as likely to report improvement in symptoms compared to those receiving a placebo, with no clear difference in minor or serious adverse effects.[151] Although there was no clear evidence of greater remission when probiotic supplements were compared with 5‐aminosalicylic acid treatment as a monotherapy, the likelihood of remission was 22% higher if probiotics were used in combination with 5-aminosalicylic acid therapy.[151]
It is unclear whether probiotics help to prevent future relapse in people with stable disease activity, either as a monotherapy or combination therapy.[152]
Fecal microbiota transplant involves the infusion of human probiotics through fecal enemas. Ulcerative colitis typically requires a more prolonged bacteriotherapy treatment than Clostridioides difficile infection does to be successful, possibly due to the time needed to heal the ulcerated epithelium. The response of ulcerative colitis is potentially very favorable with one study reporting 67.7% of people experiencing complete remission.[153] Other studies found a benefit from using fecal microbiota transplantation.[154][155][156]
Alternative medicine
[edit]A variety of alternative medicine therapies have been used for ulcerative colitis, with inconsistent results. Curcumin (turmeric) therapy, in conjunction with taking the medications mesalamine or sulfasalazine, may be effective and safe for maintaining remission in people with quiescent ulcerative colitis.[157][158] The effect of curcumin therapy alone on quiescent ulcerative colitis is unknown.[158]
Treatments using cannabis or cannabis oil are uncertain. So far, studies have not determined its effectiveness and safety.[159]
Abdominal pain management
[edit]Many interventions have been considered to manage abdominal pain in people with ulcerative colitis, including FODMAPs diet, relaxation training, yoga, kefir diet and stellate ganglion block treatment. It is unclear whether any of these are safe or effective at improving pain or reducing anxiety and depression.[160]
Nutrition
[edit]Diet can play a role in symptoms of patients with ulcerative colitis.[161]
The most avoided foods by patients are spicy foods, dairy products, alcohol, fruits and vegetables and carbonated beverages; these foods are mainly avoided during remission and to prevent relapse. In some cases, especially in the flares period, the dietary restrictions of these patients can be very severe and can lead to a compromised nutritional state. Some patients tend to eliminate gluten spontaneously, despite not having a definite diagnosis of Coeliac disease, because they believe that gluten can exacerbate gastrointestinal symptoms.[162]
Mental health
[edit]Many studies found that patients with IBD reported a higher frequency of depressive and anxiety disorders than the general population, and most studies confirm that women with IBD are more likely than men to develop affective disorders and show that up to 65% of them may have depression disorder and anxiety disorder.[163][164]
A meta analysis of interventions to improve mood (including talking therapy, antidepressants, and exercise) in people with inflammatory bowel disease found that they reduced inflammatory markers such as C-reactive protein and faecal calprotectin. Psychological therapies reduced inflammation more than antidepressants or exercise.[165][166]
Prognosis
[edit]Poor prognostic factors include: age < 40 years upon diagnosis, extensive colitis, severe colitis on endoscopy, prior hospitalization, elevated CRP and low serum albumin.[19]
Progression or remission
[edit]People with ulcerative colitis usually have an intermittent course, with periods of disease inactivity alternating with "flares" of disease. People with proctitis or left-sided colitis usually have a more benign course: only 15% progress proximally with their disease, and up to 20% can have sustained remission in the absence of any therapy. A subset of people experience a course of disease progress rapidly. In these cases, there is usually a failure to respond to medication and surgery often is performed within the first few years of disease onset.[167][168] People with more extensive disease are less likely to sustain remission, but the rate of remission is independent of the severity of the disease.[169] Several risk factors are associated with eventual need for colectomy, including: prior hospitalization for UC, extensive colitis, need for systemic steroids, young age at diagnosis, low serum albumin, elevated inflammatory markers (CRP & ESR), and severe inflammation seen during colonoscopy.[97][19] Surgical removal of the large intestine is necessary in some cases.[19]
Colorectal cancer
[edit]The risk of colorectal cancer is significantly increased in people with ulcerative colitis after ten years if involvement is beyond the splenic flexure. People with backwash ileitis might have an increased risk for colorectal carcinoma.[170] Those people with only proctitis usually have no increased risk.[19] It is recommended that people have screening colonoscopies with random biopsies to look for dysplasia after eight years of disease activity, at one to two year intervals.[171]
Mortality
[edit]People with ulcerative colitis are at similar[172] or perhaps slightly increased overall risk of death compared with the background population.[173] However, the distribution of causes-of-death differs from the general population.[172] Specific risk factors may predict worse outcomes and a higher risk of mortality in people with ulcerative colitis, including C. difficile infection[19] and cytomegalovirus infection (due to reactivation).[174]
Epidemiology
[edit]Together with Crohn's disease, about 11.2 million people were affected as of 2015[update].[175] Each year it newly occurs in 1 to 20 per 100,000 people, and 5 to 500 per 100,000 individuals are affected.[7][9] The disease is more common in North America and Europe than other regions.[9] Often it begins in people aged 15 to 30 years, or among those over 60.[1] Males and females appear to be affected in equal proportions.[7] It has also become more common since the 1950s.[7][9] Together, ulcerative colitis and Crohn's disease affect about a million people in the United States.[176] With appropriate treatment the risk of death appears the same as that of the general population.[3] The first description of ulcerative colitis occurred around the 1850s.[9]
Each year, ulcerative colitis newly occurs in 1 to 20 per 100,000 people (incidence), and there are a total of 5–500 per 100,000 individuals with the disease (prevalence).[7][9] In 2015, a worldwide total of 47,400 people died due to inflammatory bowel disease (UC and Crohn's disease).[6] The peak onset is between 30 and 40 years of age,[12] with a second peak of onset occurring in the 6th decade of life.[177] Ulcerative colitis is equally common among men and women.[12][7] With appropriate treatment the risk of death appears similar to that of the general population.[3] UC has become more common since the 1950s.[7][9]
The geographic distribution of UC and Crohn's disease is similar worldwide,[178] with the highest number of new cases a year of UC found in Canada, New Zealand and the United Kingdom.[179] The disease is more common in North America and Europe than other regions.[9] In general, higher rates are seen in northern locations compared to southern locations in Europe[180] and the United States.[181] UC is more common in western Europe compared with eastern Europe.[182] Worldwide, the prevalence of UC varies from 2 to 299 per 100,000 people.[5] Together, ulcerative colitis and Crohn's disease affect about a million people in the United States.[176]
As with Crohn's disease, the rates of UC are greater among Ashkenazi Jews and decreases progressively in other persons of Jewish descent, non-Jewish Caucasians, Africans, Hispanics, and Asians.[22] Appendectomy prior to age 20 for appendicitis[183] and current tobacco use[184] are protective against development of UC.[12] However, former tobacco use is associated with a higher risk of developing the disease.[184][12]
United States
[edit]As of 2004[update], the number of new cases of UC in the United States was between 2.2 and 14.3 per 100,000 per year.[185] The number of people affected in the United States in 2004 was between 37 and 246 per 100,000.[185]
Canada
[edit]In Canada, between 1998 and 2000, the number of new cases per year was 12.9 per 100,000 population or 4,500 new cases. The number of people affected was estimated to be 211 per 100,000 or 104,000.[186]
United Kingdom
[edit]In the United Kingdom 10 per 100,000 people newly develop the condition a year while the number of people affected is 243 per 100,000. Approximately 146,000 people in the United Kingdom have been diagnosed with UC.[187]
History
[edit]The term ulcerative colitis was first used by Samuel Wilks in 1859. The term entered general medical vocabulary afterwards in 1888 with William Hale-White publishing a report of various cases of "ulcerative colitis".[188]
UC was the first subtype of IBD to be identified.[188]
Research
[edit]Helminthic therapy using the whipworm Trichuris suis has been shown in a randomized control trial from Iowa to show benefit in people with ulcerative colitis.[189] The therapy tests the hygiene hypothesis which argues that the absence of helminths in the colons of people in the developed world may lead to inflammation. Both helminthic therapy and fecal microbiota transplant induce a characteristic Th2 white cell response in the diseased areas, which was unexpected given that ulcerative colitis was thought to involve Th2 overproduction.[189]
Alicaforsen is a first generation antisense oligodeoxynucleotide designed to bind specifically to the human ICAM-1 messenger RNA through Watson-Crick base pair interactions in order to subdue expression of ICAM-1.[190] ICAM-1 propagates an inflammatory response promoting the extravasation and activation of leukocytes (white blood cells) into inflamed tissue.[190] Increased expression of ICAM-1 has been observed within the inflamed intestinal mucosa of ulcerative colitis patients, where ICAM-1 over production correlated with disease activity.[191] This suggests that ICAM-1 is a potential therapeutic target in the treatment of ulcerative colitis.[192]
Gram positive bacteria present in the lumen could be associated with extending the time of relapse for ulcerative colitis.[193]
A series of drugs in development looks to disrupt the inflammation process by selectively targeting an ion channel in the inflammation signaling cascade known as KCa3.1.[194] In a preclinical study in rats and mice, inhibition of KCa3.1 disrupted the production of Th1 cytokines IL-2 and TNF-α and decreased colon inflammation as effectively as sulfasalazine.[194]
Neutrophil extracellular traps[195] and the resulting degradation of the extracellular matrix[196] have been reported in the colon mucosa in ulcerative colitis patients in clinical remission, indicating the involvement of the innate immune system in the etiology.[195]
Fexofenadine, an antihistamine drug used in treatment of allergies, has shown promise in a combination therapy in some studies.[197][198] Opportunely, low gastrointestinal absorption (or high absorbed drug gastrointestinal secretion) of fexofenadine results in higher concentration at the site of inflammation. Thus, the drug may locally decrease histamine secretion by involved gastrointestinal mast cells and alleviate the inflammation.[198]
There is evidence that etrolizumab is effective for ulcerative colitis, with phase 3 trials underway as of 2016.[8][199][200][201] Etrolizumab is a humanized monoclonal antibody that targets the β7 subunit of integrins α4β7 and αEβ7, ultimately blocking migration and retention of leukocytes in the intestinal mucosa.[200] As of early 2022, Roche halted clinical trials for the use of etrolizumab in the treatment of ulcerative colitis.[202]
A type of leukocyte apheresis, known as granulocyte and monocyte adsorptive apheresis, still requires large-scale trials to determine whether or not it is effective.[203] Results from small trials have been tentatively positive.[204]
Notable cases
[edit]References
[edit]- ^ a b c d e f g h i j k l m n o p q r s t u v w "Ulcerative Colitis". NIDDK. September 2014. Retrieved 3 August 2016.
- ^ "Ulcerative Colitis". Autoimmune Registry Inc. Retrieved 15 June 2022.[permanent dead link]
- ^ a b c d Wanderås MH, Moum BA, Høivik ML, Hovde Ø (May 2016). "Predictive factors for a severe clinical course in ulcerative colitis: Results from population-based studies". World Journal of Gastrointestinal Pharmacology and Therapeutics. 7 (2): 235–241. doi:10.4292/wjgpt.v7.i2.235. PMC 4848246. PMID 27158539.
- ^ Runge MS, Greganti MA (2008). Netter's Internal Medicine E-Book. Elsevier Health Sciences. p. 428. ISBN 9781437727722.
- ^ a b Molodecky NA, Soon IS, Rabi DM, Ghali WA, Ferris M, Chernoff G, et al. (January 2012). "Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review". Gastroenterology. 142 (1): 46–54.e42, quiz e30. doi:10.1053/j.gastro.2011.10.001. PMID 22001864.
- ^ a b Wang H, Naghavi M, Allen C, Barber RM, Bhutta ZA, Carter A, et al. (GBD 2015 Mortality and Causes of Death Collaborators) (October 2016). "Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1459–1544. doi:10.1016/s0140-6736(16)31012-1. PMC 5388903. PMID 27733281.
- ^ a b c d e f g h Ford AC, Moayyedi P, Hanauer SB (February 2013). "Ulcerative colitis". BMJ. 346: f432. doi:10.1136/bmj.f432. PMID 23386404. S2CID 14778938.
- ^ a b c Akiho H, Yokoyama A, Abe S, Nakazono Y, Murakami M, Otsuka Y, et al. (November 2015). "Promising biological therapies for ulcerative colitis: A review of the literature". World Journal of Gastrointestinal Pathophysiology. 6 (4): 219–227. doi:10.4291/wjgp.v6.i4.219. PMC 4644886. PMID 26600980.
- ^ a b c d e f g h i j Danese S, Fiocchi C (November 2011). "Ulcerative colitis". The New England Journal of Medicine. 365 (18): 1713–1725. doi:10.1056/NEJMra1102942. PMID 22047562.
- ^ a b c d e f "Inflammatorisk tarmsjukdom, kronisk, IBD". internetmedicin.se (in Swedish). 4 January 2009. Archived from the original on 20 August 2010.
- ^ Hanauer SB, Sandborn W (March 2001). "Management of Crohn's disease in adults". The American Journal of Gastroenterology. 96 (3): 635–43. doi:10.1111/j.1572-0241.2001.3671_c.x (inactive 2 November 2024). PMID 11280528. S2CID 31219115.
{{cite journal}}: CS1 maint: DOI inactive as of November 2024 (link) - ^ a b c d e f g h i j k l m n o p Ungaro R, Mehandru S, Allen PB, Peyrin-Biroulet L, Colombel JF (April 2017). "Ulcerative colitis". Lancet. 389 (10080): 1756–1770. doi:10.1016/S0140-6736(16)32126-2. PMC 6487890. PMID 27914657.
- ^ a b c d e f g h i j k l m n o p Gros B, Kaplan GG (12 September 2023). "Ulcerative Colitis in Adults: A Review". JAMA. 330 (10): 951–965. doi:10.1001/jama.2023.15389. PMID 37698559. S2CID 261696018.
- ^ a b c d e Magro F, Gionchetti P, Eliakim R, Ardizzone S, Armuzzi A, Barreiro-de Acosta M, et al. (June 2017). "Third European Evidence-based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 1: Definitions, Diagnosis, Extra-intestinal Manifestations, Pregnancy, Cancer Surveillance, Surgery, and Ileo-anal Pouch Disorders". Journal of Crohn's & Colitis. 11 (6): 649–670. doi:10.1093/ecco-jcc/jjx008. PMID 28158501.
- ^ Kaitha S, Bashir M, Ali T (August 2015). "Iron deficiency anemia in inflammatory bowel disease". World Journal of Gastrointestinal Pathophysiology. 6 (3): 62–72. doi:10.4291/wjgp.v6.i3.62. PMC 4540708. PMID 26301120.
- ^ Hanauer SB (March 1996). "Inflammatory bowel disease". The New England Journal of Medicine. 334 (13): 841–848. doi:10.1056/NEJM199603283341307. PMID 8596552.
- ^ Rosenberg L, Lawlor GO, Zenlea T, Goldsmith JD, Gifford A, Falchuk KR, et al. (2013). "Predictors of endoscopic inflammation in patients with ulcerative colitis in clinical remission". Inflammatory Bowel Diseases. 19 (4): 779–784. doi:10.1097/MIB.0b013e3182802b0e. PMC 3749843. PMID 23446338.
- ^ a b c d e f Colìa R, Corrado A, Cantatore FP (December 2016). "Rheumatologic and extraintestinal manifestations of inflammatory bowel diseases". Annals of Medicine. 48 (8): 577–585. doi:10.1080/07853890.2016.1195011. PMID 27310096. S2CID 1796160.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x Rubin DT, Ananthakrishnan AN, Siegel CA, Sauer BG, Long MD (March 2019). "ACG Clinical Guideline: Ulcerative Colitis in Adults". The American Journal of Gastroenterology. 114 (3): 384–413. doi:10.14309/ajg.0000000000000152. PMID 30840605. S2CID 73473272.
- ^ Feuerstein JD, Cheifetz AS (July 2017). "Crohn Disease: Epidemiology, Diagnosis, and Management". Mayo Clinic Proceedings. 92 (7): 1088–1103. doi:10.1016/j.mayocp.2017.04.010. PMID 28601423. S2CID 20223406.
- ^ Haskell H, Andrews CW, Reddy SI, Dendrinos K, Farraye FA, Stucchi AF, et al. (November 2005). "Pathologic features and clinical significance of "backwash" ileitis in ulcerative colitis". The American Journal of Surgical Pathology. 29 (11): 1472–1481. doi:10.1097/01.pas.0000176435.19197.88. PMID 16224214. S2CID 42108108.
- ^ a b Fauci et al. Harrison's Internal Medicine, 17th ed. New York: McGraw-Hill Medical, 2008. ISBN 978-0-07-159991-7
- ^ a b "UpToDate". www.uptodate.com. Retrieved 8 November 2022.
- ^ "UpToDate". www.uptodate.com. Retrieved 9 November 2022.
- ^ Greenstein AJ, Janowitz HD, Sachar DB (September 1976). "The extra-intestinal complications of Crohn's disease and ulcerative colitis: a study of 700 patients". Medicine. 55 (5): 401–412. doi:10.1097/00005792-197609000-00004. PMID 957999.
- ^ Bernstein CN, Blanchard JF, Rawsthorne P, Yu N (April 2001). "The prevalence of extraintestinal diseases in inflammatory bowel disease: a population-based study". The American Journal of Gastroenterology. 96 (4): 1116–1122. doi:10.1111/j.1572-0241.2001.03756.x. PMID 11316157.
- ^ Harbord M, Annese V, Vavricka SR, Allez M, Barreiro-de Acosta M, Boberg KM, et al. (March 2016). "The First European Evidence-based Consensus on Extra-intestinal Manifestations in Inflammatory Bowel Disease". Journal of Crohn's & Colitis. 10 (3): 239–254. doi:10.1093/ecco-jcc/jjv213. PMID 26614685.
- ^ a b c d e f g h i j k l m Feuerstein JD, Moss AC, Farraye FA (July 2019). "Ulcerative Colitis". Mayo Clinic Proceedings. 94 (7): 1357–1373. doi:10.1016/j.mayocp.2019.01.018. PMID 31272578.
- ^ Langan RC, Gotsch PB, Krafczyk MA, Skillinge DD (November 2007). "Ulcerative colitis: diagnosis and treatment". American Family Physician. 76 (9): 1323–1330. PMID 18019875.
- ^ Vavricka SR, Schoepfer A, Scharl M, Lakatos PL, Navarini A, Rogler G (August 2015). "Extraintestinal Manifestations of Inflammatory Bowel Disease". Inflammatory Bowel Diseases. 21 (8): 1982–1992. doi:10.1097/MIB.0000000000000392. PMC 4511685. PMID 26154136.
- ^ a b c d Muhvić-Urek M, Tomac-Stojmenović M, Mijandrušić-Sinčić B (July 2016). "Oral pathology in inflammatory bowel disease". World Journal of Gastroenterology. 22 (25): 5655–5667. doi:10.3748/wjg.v22.i25.5655. PMC 4932203. PMID 27433081.
- ^ a b c Troncoso LL, Biancardi AL, de Moraes HV, Zaltman C (August 2017). "Ophthalmic manifestations in patients with inflammatory bowel disease: A review". World Journal of Gastroenterology. 23 (32): 5836–5848. doi:10.3748/wjg.v23.i32.5836. PMC 5583569. PMID 28932076.
- ^ Schonberg S, Stokkermans TJ (January 2020). Episcleritis. StatPearls. PMID 30521217.
- ^ a b c d e f g Langholz E (March 2010). "Current trends in inflammatory bowel disease: the natural history". Therapeutic Advances in Gastroenterology. 3 (2): 77–86. doi:10.1177/1756283X10361304. PMC 3002570. PMID 21180592.
- ^ a b Farhi D, Cosnes J, Zizi N, Chosidow O, Seksik P, Beaugerie L, et al. (September 2008). "Significance of erythema nodosum and pyoderma gangrenosum in inflammatory bowel diseases: a cohort study of 2402 patients". Medicine. 87 (5): 281–293. doi:10.1097/MD.0b013e318187cc9c. ISSN 1536-5964. PMID 18794711. S2CID 6905740.
- ^ Chen WT, Chi CC (1 September 2019). "Association of Hidradenitis Suppurativa With Inflammatory Bowel Disease: A Systematic Review and Meta-analysis". JAMA Dermatology. 155 (9): 1022–1027. doi:10.1001/jamadermatol.2019.0891. ISSN 2168-6084. PMC 6625071. PMID 31290938.
- ^ a b c Cheng K, Faye AS (March 2020). "Venous thromboembolism in inflammatory bowel disease". World Journal of Gastroenterology. 26 (12): 1231–1241. doi:10.3748/wjg.v26.i12.1231. PMC 7109271. PMID 32256013. S2CID 214946656.
- ^ a b Nguyen GC, Bernstein CN, Bitton A, Chan AK, Griffiths AM, Leontiadis GI, et al. (March 2014). "Consensus statements on the risk, prevention, and treatment of venous thromboembolism in inflammatory bowel disease: Canadian Association of Gastroenterology". Gastroenterology. 146 (3): 835–848.e6. doi:10.1053/j.gastro.2014.01.042. PMID 24462530.
- ^ Andrade AR, Barros LL, Azevedo MF, Carlos AS, Damião AO, Sipahi AM, et al. (April 2018). "Risk of thrombosis and mortality in inflammatory bowel disease". Clinical and Translational Gastroenterology. 9 (4): 142. doi:10.1038/s41424-018-0013-8. PMC 5886983. PMID 29618721.
- ^ Olsson R, Danielsson A, Järnerot G, Lindström E, Lööf L, Rolny P, et al. (May 1991). "Prevalence of primary sclerosing cholangitis in patients with ulcerative colitis". Gastroenterology. 100 (5 Pt 1): 1319–1323. doi:10.1016/0016-5085(91)90784-I. PMID 2013375.
- ^ Rasmussen HH, Fallingborg JF, Mortensen PB, Vyberg M, Tage-Jensen U, Rasmussen SN (June 1997). "Hepatobiliary dysfunction and primary sclerosing cholangitis in patients with Crohn's disease". Scandinavian Journal of Gastroenterology. 32 (6): 604–610. doi:10.3109/00365529709025107. ISSN 0036-5521. PMID 9200295.
- ^ Nachimuthu S. "Crohn's disease". eMedicineHealth. Archived from the original on 9 December 2019. Retrieved 8 December 2019.
- ^ Ko IK, Kim BG, Awadallah A, Mikulan J, Lin P, Letterio JJ, et al. (July 2010). "Targeting improves MSC treatment of inflammatory bowel disease". Molecular Therapy. 18 (7): 1365–1372. doi:10.1038/mt.2010.54. PMC 2911249. PMID 20389289.
- ^ a b Orholm M, Binder V, Sørensen TI, Rasmussen LP, Kyvik KO (October 2000). "Concordance of inflammatory bowel disease among Danish twins. Results of a nationwide study". Scandinavian Journal of Gastroenterology. 35 (10): 1075–1081. doi:10.1080/003655200451207. PMID 11099061. S2CID 218907577.
- ^ Tysk C, Lindberg E, Järnerot G, Flodérus-Myrhed B (July 1988). "Ulcerative colitis and Crohn's disease in an unselected population of monozygotic and dizygotic twins. A study of heritability and the influence of smoking". Gut. 29 (7): 990–996. doi:10.1136/gut.29.7.990. PMC 1433769. PMID 3396969.
- ^ a b c Baumgart DC, Sandborn WJ (May 2007). "Inflammatory bowel disease: clinical aspects and established and evolving therapies". Lancet. 369 (9573): 1641–57. doi:10.1016/S0140-6736(07)60751-X. PMID 17499606. S2CID 35264387.
- ^ Cho JH, Nicolae DL, Ramos R, Fields CT, Rabenau K, Corradino S, et al. (May 2000). "Linkage and linkage disequilibrium in chromosome band 1p36 in American Chaldeans with inflammatory bowel disease". Human Molecular Genetics. 9 (9): 1425–1432. doi:10.1093/hmg/9.9.1425. PMID 10814724.
- ^ Shah A, Walker M, Burger D, Martin N, von Wulffen M, Koloski N, et al. (August 2019). "Link Between Celiac Disease and Inflammatory Bowel Disease". Journal of Clinical Gastroenterology. 53 (7): 514–522. doi:10.1097/MCG.0000000000001033. PMID 29762265. S2CID 44102071.
- ^ Fu Y, Lee CH, Chi CC (December 2018). "Association of Psoriasis With Inflammatory Bowel Disease: A Systematic Review and Meta-analysis". JAMA Dermatology. 154 (12): 1417–1423. doi:10.1001/jamadermatol.2018.3631. PMC 6583370. PMID 30422277.
- ^ Katsanos KH, Voulgari PV, Tsianos EV (August 2012). "Inflammatory bowel disease and lupus: a systematic review of the literature". Journal of Crohn's & Colitis. 6 (7): 735–742. doi:10.1016/j.crohns.2012.03.005. PMID 22504032.
- ^ Chen Y, Chen L, Xing C, Deng G, Zeng F, Xie T, et al. (June 2020). "The risk of rheumatoid arthritis among patients with inflammatory bowel disease: a systematic review and meta-analysis". BMC Gastroenterology. 20 (1): 192. doi:10.1186/s12876-020-01339-3. PMC 7301504. PMID 32552882.
- ^ Sieg I, Beckh K, Kersten U, Doss MO (November 1991). "Manifestation of acute intermittent porphyria in patients with chronic inflammatory bowel disease". Zeitschrift für Gastroenterologie. 29 (11): 602–605. PMID 1771936.
- ^ Xu L, Lochhead P, Ko Y, Claggett B, Leong RW, Ananthakrishnan AN (November 2017). "Systematic review with meta-analysis: breastfeeding and the risk of Crohn's disease and ulcerative colitis". Alimentary Pharmacology & Therapeutics. 46 (9): 780–789. doi:10.1111/apt.14291. PMC 5688338. PMID 28892171.
- ^ Corrao G, Tragnone A, Caprilli R, Trallori G, Papi C, Andreoli A, et al. (June 1998). "Risk of inflammatory bowel disease attributable to smoking, oral contraception and breastfeeding in Italy: a nationwide case-control study. Cooperative Investigators of the Italian Group for the Study of the Colon and the Rectum (GISC)". International Journal of Epidemiology. 27 (3): 397–404. doi:10.1093/ije/27.3.397. PMID 9698126.
- ^ Wolverton SE, Harper JC (April 2013). "Important controversies associated with isotretinoin therapy for acne". American Journal of Clinical Dermatology. 14 (2): 71–76. doi:10.1007/s40257-013-0014-z. PMID 23559397. S2CID 918753.
- ^ Järnerot G, Järnmark I, Nilsson K (November 1983). "Consumption of refined sugar by patients with Crohn's disease, ulcerative colitis, or irritable bowel syndrome". Scandinavian Journal of Gastroenterology. 18 (8): 999–1002. doi:10.3109/00365528309181832. PMID 6673083.
- ^ Geerling BJ, Dagnelie PC, Badart-Smook A, Russel MG, Stockbrügger RW, Brummer RJ (April 2000). "Diet as a risk factor for the development of ulcerative colitis". The American Journal of Gastroenterology. 95 (4): 1008–1013. doi:10.1111/j.1572-0241.2000.01942.x. PMID 10763951. S2CID 11295804.
- ^ Jowett SL, Seal CJ, Pearce MS, Phillips E, Gregory W, Barton JR, et al. (October 2004). "Influence of dietary factors on the clinical course of ulcerative colitis: a prospective cohort study". Gut. 53 (10): 1479–1484. doi:10.1136/gut.2003.024828. PMC 1774231. PMID 15361498.
- ^ Andersen V, Olsen A, Carbonnel F, Tjønneland A, Vogel U (March 2012). "Diet and risk of inflammatory bowel disease". Digestive and Liver Disease. 44 (3): 185–194. doi:10.1016/j.dld.2011.10.001. PMID 22055893.
- ^ Tilg H, Kaser A (October 2004). "Diet and relapsing ulcerative colitis: take off the meat?". Gut. 53 (10): 1399–1401. doi:10.1136/gut.2003.035287. PMC 1774255. PMID 15361484.
- ^ Moore J, Babidge W, Millard S, Roediger W (January 1998). "Colonic luminal hydrogen sulfide is not elevated in ulcerative colitis". Digestive Diseases and Sciences. 43 (1): 162–165. doi:10.1023/A:1018848709769. PMID 9508519. S2CID 20919357.
- ^ Jørgensen J, Mortensen PB (August 2001). "Hydrogen sulfide and colonic epithelial metabolism: implications for ulcerative colitis". Digestive Diseases and Sciences. 46 (8): 1722–1732. doi:10.1023/A:1010661706385. PMID 11508674. S2CID 30373968.
- ^ Picton R, Eggo MC, Langman MJ, Singh S (February 2007). "Impaired detoxication of hydrogen sulfide in ulcerative colitis?". Digestive Diseases and Sciences. 52 (2): 373–378. doi:10.1007/s10620-006-9529-y. PMID 17216575. S2CID 22547709.
- ^ Nicole W (2013). "PFOA and Cancer in a Highly Exposed Community: New Findings from the C8 Science Panel". Environmental Health Perspectives. 121 (11–12): A340. doi:10.1289/ehp.121-A340. PMC 3855507. PMID 24284021.
- ^ a b Roediger WE, Moore J, Babidge W (August 1997). "Colonic sulfide in pathogenesis and treatment of ulcerative colitis". Digestive Diseases and Sciences. 42 (8): 1571–1579. doi:10.1023/A:1018851723920. PMID 9286219. S2CID 25496705.
- ^ Pierce ES (2018). "Could Mycobacterium avium subspecies paratuberculosis cause Crohn's disease, ulcerative colitis…and colorectal cancer?". Infectious Agents and Cancer. 13: 1. doi:10.1186/s13027-017-0172-3. PMC 5753485. PMID 29308085.
- ^ Elson CO, Cong Y, Weaver CT, Schoeb TR, McClanahan TK, Fick RB, et al. (2007). "Monoclonal anti-interleukin 23 reverses active colitis in a T cell-mediated model in mice". Gastroenterology. 132 (7): 2359–70. doi:10.1053/j.gastro.2007.03.104. PMID 17570211.
- ^ Levine J, Ellis CJ, Furne JK, Springfield J, Levitt MD (January 1998). "Fecal hydrogen sulfide production in ulcerative colitis". The American Journal of Gastroenterology. 93 (1): 83–87. doi:10.1111/j.1572-0241.1998.083_c.x. PMID 9448181. S2CID 3141574.
- ^ a b Dassopoulos T, Cohen RD, Scherl EJ, Schwartz RM, Kosinski L, Regueiro MD (July 2015). "Ulcerative Colitis Care Pathway". Gastroenterology. 149 (1): 238–245. doi:10.1053/j.gastro.2015.05.036. PMID 26025078.
- ^ a b c d e Nikolaus S, Schreiber S (November 2007). "Diagnostics of inflammatory bowel disease". Gastroenterology. 133 (5): 1670–1689. doi:10.1053/j.gastro.2007.09.001. ISSN 1528-0012. PMID 17983810.
- ^ Ulcerative colitis at eMedicine
- ^ Walmsley RS, Ayres RC, Pounder RE, Allan RN (July 1998). "A simple clinical colitis activity index". Gut. 43 (1): 29–32. doi:10.1136/gut.43.1.29. PMC 1727189. PMID 9771402.
- ^ a b Lamb CA, Kennedy NA, Raine T, Hendy PA, Smith PJ, Limdi JK, et al. (December 2019). "British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults". Gut. 68 (Suppl 3): s1–s106. doi:10.1136/gutjnl-2019-318484. PMC 6872448. PMID 31562236.
- ^ Politis DS, Papamichael K, Katsanos KH, Koulouridis I, Mavromati D, Tsianos EV, et al. (March 2019). "Presence of pseudopolyps in ulcerative colitis is associated with a higher risk for treatment escalation". Annals of Gastroenterology. 32 (2): 168–173. doi:10.20524/aog.2019.0357. PMC 6394261. PMID 30837789.
- ^ "Azathioprine Product Information" (PDF). Access FDA. Food and Drug Administration.
- ^ Dignass AU, Gasche C, Bettenworth D, Birgegård G, Danese S, Gisbert JP, et al. (March 2015). "European consensus on the diagnosis and management of iron deficiency and anaemia in inflammatory bowel diseases". Journal of Crohn's & Colitis. 9 (3): 211–222. doi:10.1093/ecco-jcc/jju009. PMID 25518052.
- ^ Del Pinto R, Pietropaoli D, Chandar AK, Ferri C, Cominelli F (November 2015). "Association Between Inflammatory Bowel Disease and Vitamin D Deficiency: A Systematic Review and Meta-analysis". Inflammatory Bowel Diseases. 21 (11): 2708–2717. doi:10.1097/MIB.0000000000000546. PMC 4615394. PMID 26348447.
- ^ Mosli MH, Zou G, Garg SK, Feagan SG, MacDonald JK, Chande N, et al. (June 2015). "C-Reactive Protein, Fecal Calprotectin, and Stool Lactoferrin for Detection of Endoscopic Activity in Symptomatic Inflammatory Bowel Disease Patients: A Systematic Review and Meta-Analysis". The American Journal of Gastroenterology. 110 (6): 802–19, quiz 820. doi:10.1038/ajg.2015.120. PMID 25964225. S2CID 26111716.
- ^ Bryant RV, Friedman AB, Wright EK, Taylor KM, Begun J, Maconi G, et al. (May 2018). "Gastrointestinal ultrasound in inflammatory bowel disease: an underused resource with potential paradigm-changing application". Gut. 67 (5): 973–985. doi:10.1136/gutjnl-2017-315655. PMID 29437914. S2CID 3344377.
- ^ Allocca M, Filippi E, Costantino A, Bonovas S, Fiorino G, Furfaro F, et al. (May 2021). "Milan ultrasound criteria are accurate in assessing disease activity in ulcerative colitis: external validation". United European Gastroenterology Journal. 9 (4): 438–442. doi:10.1177/2050640620980203. PMC 8259285. PMID 33349199.
- ^ Allocca M, Fiorino G, Bonovas S, Furfaro F, Gilardi D, Argollo M, et al. (28 November 2018). "Accuracy of Humanitas Ultrasound Criteria in Assessing Disease Activity and Severity in Ulcerative Colitis: A Prospective Study". Journal of Crohn's and Colitis. 12 (12): 1385–1391. doi:10.1093/ecco-jcc/jjy107. PMC 6260119. PMID 30085066.
- ^ Shirley DA, Moonah S (July 2016). "Fulminant Amebic Colitis after Corticosteroid Therapy: A Systematic Review". PLOS Neglected Tropical Diseases. 10 (7): e0004879. doi:10.1371/journal.pntd.0004879. PMC 4965027. PMID 27467600.
- ^ Tremaine WJ (April 2012). "Is indeterminate colitis determinable?". Current Gastroenterology Reports. 14 (2): 162–165. doi:10.1007/s11894-012-0244-x. PMID 22314810. S2CID 40346031.
- ^ Kim B, Barnett JL, Kleer CG, Appelman HD (November 1999). "Endoscopic and histological patchiness in treated ulcerative colitis". The American Journal of Gastroenterology. 94 (11): 3258–3262. doi:10.1111/j.1572-0241.1999.01533.x. hdl:2027.42/74642. ISSN 0002-9270. PMID 10566726. S2CID 11446833.
- ^ Woodruff JM, Hansen JA, Good RA, Santos GW, Slavin RE (December 1976). "The pathology of the graft-versus-host reaction (GVHR) in adults receiving bone marrow transplants". Transplantation Proceedings. 8 (4): 675–684. ISSN 0041-1345. PMID 11596.
- ^ a b c Hanauer SB, Sandborn W (March 2001). "Management of Crohn's disease in adults". The American Journal of Gastroenterology. 96 (3): 635–43. doi:10.1111/j.1572-0241.2001.3671_c.x (inactive 2 November 2024). PMID 11280528. S2CID 31219115.
{{cite journal}}: CS1 maint: DOI inactive as of November 2024 (link) - ^ Broomé U, Bergquist A (February 2006). "Primary sclerosing cholangitis, inflammatory bowel disease, and colon cancer". Seminars in Liver Disease. 26 (1): 31–41. doi:10.1055/s-2006-933561. PMID 16496231. S2CID 260320940.
- ^ Shepherd NA (August 2002). "Granulomas in the diagnosis of intestinal Crohn's disease: a myth exploded?". Histopathology. 41 (2): 166–8. doi:10.1046/j.1365-2559.2002.01441.x. PMID 12147095. S2CID 36907992.
- ^ Mahadeva U, Martin JP, Patel NK, Price AB (July 2002). "Granulomatous ulcerative colitis: a re-appraisal of the mucosal granuloma in the distinction of Crohn's disease from ulcerative colitis". Histopathology. 41 (1): 50–5. doi:10.1046/j.1365-2559.2002.01416.x. PMID 12121237. S2CID 29476514.
- ^ DeRoche TC, Xiao SY, Liu X (August 2014). "Histological evaluation in ulcerative colitis". Gastroenterology Report. 2 (3): 178–92. doi:10.1093/gastro/gou031. PMC 4124271. PMID 24942757.
- ^ Chen JH, Andrews JM, Kariyawasam V, Moran N, Gounder P, Collins G, et al. (July 2016). "Review article: acute severe ulcerative colitis - evidence-based consensus statements". Alimentary Pharmacology & Therapeutics. 44 (2): 127–144. doi:10.1111/apt.13670. PMID 27226344.
- ^ "Should You Try a Low-Residue Diet?". WebMD. 25 October 2016. Retrieved 29 April 2017.
- ^ Manual of Clinical Nutrition Management (PDF). Compass Group. 2013.
- ^ Roncoroni L, Gori R, Elli L, et al. Nutrition in Patients with Inflammatory Bowel Diseases: A Narrative Review. Nutrients. 2022;14(4):751. Published 2022 Feb 10. doi:10.3390/nu14040751
- ^ a b Ham M, Moss AC (2012). "Mesalamine in the treatment and maintenance of remission of ulcerative colitis". Expert Review of Clinical Pharmacology. 5 (2): 113–123. doi:10.1586/ecp.12.2. ISSN 1751-2433. PMC 3314328. PMID 22390554.
- ^ a b Raine T, Bonovas S, Burisch J, Kucharzik T, Adamina M, Annese V, et al. (28 January 2022). "ECCO Guidelines on Therapeutics in Ulcerative Colitis: Medical Treatment". Journal of Crohn's and Colitis. 16 (1): 2–17. doi:10.1093/ecco-jcc/jjab178. ISSN 1873-9946. PMID 34635919.
- ^ a b c Feuerstein JD, Isaacs KL, Schneider Y, Siddique SM, Falck-Ytter Y, Singh S (April 2020). "AGA Clinical Practice Guidelines on the Management of Moderate to Severe Ulcerative Colitis". Gastroenterology. 158 (5): 1450–1461. doi:10.1053/j.gastro.2020.01.006. PMC 7175923. PMID 31945371.
- ^ "Skyrizi- risankizumab-rzaa kit". DailyMed. 18 June 2024. Retrieved 2 November 2024.
- ^ Sandborn W (2002). "Rational selection of oral 5-aminosalicylate formulations and prodrugs for the treatment of ulcerative colitis". The American Journal of Gastroenterology. 97 (12): 2939–2941. doi:10.1016/S0002-9270(02)05509-0. PMID 12492172.
- ^ Ko CW, Singh S, Feuerstein JD, Falck-Ytter C, Falck-Ytter Y, Cross RK, et al. (2019). "AGA Clinical Practice Guidelines on the Management of Mild-to-Moderate Ulcerative Colitis". Gastroenterology. 156 (3): 748–764. doi:10.1053/j.gastro.2018.12.009. PMC 6858922. PMID 30576644.
- ^ "FDA approves Uceris as ulcerative colitis treatment". Healio Gastroenterology. 15 January 2013.
- ^ "UCERIS (budesonide) extended release tablets label" (PDF). FDA.
- ^ "FDA approves new treatment for moderately to severely active ulcerative colitis". U.S. Food and Drug Administration (FDA) (Press release). 30 May 2018. Retrieved 31 May 2018.
- ^ Chande N, Wang Y, MacDonald JK, McDonald JW (August 2014). "Methotrexate for induction of remission in ulcerative colitis". The Cochrane Database of Systematic Reviews. 8 (8): CD006618. doi:10.1002/14651858.CD006618.pub3. PMC 6486224. PMID 25162749.
- ^ Krishnamoorthy R, Abrams KR, Guthrie N, Samuel S, Thomas T (28 May 2012). "PWE-237 Ciclosporin in acute severe ulcerative colitis: a meta-analysis". Gut. 61 (Suppl 2): A394.2–A394. doi:10.1136/gutjnl-2012-302514d.237. S2CID 74798482.
- ^ Ogata H, Kato J, Hirai F, Hida N, Matsui T, Matsumoto T, et al. (May 2012). "Double-blind, placebo-controlled trial of oral tacrolimus (FK506) in the management of hospitalized patients with steroid-refractory ulcerative colitis". Inflammatory Bowel Diseases. 18 (5): 803–808. doi:10.1002/ibd.21853. PMID 21887732. S2CID 1294555.
- ^ Lichtiger S, Present DH, Kornbluth A, Gelernt I, Bauer J, Galler G, et al. (June 1994). "Cyclosporine in severe ulcerative colitis refractory to steroid therapy". The New England Journal of Medicine. 330 (26): 1841–1845. doi:10.1056/NEJM199406303302601. PMID 8196726.
- ^ Weisshof R, Ollech JE, El Jurdi K, Yvellez OV, Cohen RD, Sakuraba A, et al. (September 2019). "Ciclosporin Therapy After Infliximab Failure in Hospitalized Patients With Acute Severe Colitis is Effective and Safe". Journal of Crohn's & Colitis. 13 (9): 1105–1110. doi:10.1093/ecco-jcc/jjz032. PMC 7327272. PMID 30726894.
- ^ "U.S. FDA Approves Pfizer's Velsipity for Adults with Moderately to Severely Active Ulcerative Colitis (UC)" (Press release). Pfizer. 13 October 2023. Retrieved 13 October 2023 – via Business Wire.
- ^ Azad Khan AK, Piris J, Truelove SC (October 1977). "An experiment to determine the active therapeutic moiety of sulphasalazine". Lancet. 2 (8044): 892–895. doi:10.1016/s0140-6736(77)90831-5. PMID 72239. S2CID 44785199.
- ^ Murray A, Nguyen TM, Parker CE, Feagan BG, MacDonald JK (August 2020). "Oral 5-aminosalicylic acid for induction of remission in ulcerative colitis". The Cochrane Database of Systematic Reviews. 2020 (8): CD000543. doi:10.1002/14651858.CD000543.pub5. PMC 8189994. PMID 32786164.
- ^ Murray A, Nguyen TM, Parker CE, Feagan BG, MacDonald JK (August 2020). "Oral 5-aminosalicylic acid for maintenance of remission in ulcerative colitis". The Cochrane Database of Systematic Reviews. 2020 (8): CD000544. doi:10.1002/14651858.CD000544.pub5. PMC 8094989. PMID 32856298.
- ^ Marshall JK, Thabane M, Steinhart AH, Newman JR, Anand A, Irvine EJ (November 2012). "Rectal 5-aminosalicylic acid for maintenance of remission in ulcerative colitis". The Cochrane Database of Systematic Reviews. 11: CD004118. doi:10.1002/14651858.CD004118.pub2. PMID 23152224.
- ^ Salahudeen MS (June 2019). "A review of current evidence allied to step-up and top-down medication therapy in inflammatory bowel disease". Drugs of Today. 55 (6): 385–405. doi:10.1358/dot.2019.55.6.2969816. PMID 31250843. S2CID 195763151.
- ^ Axelrad JE, Lichtiger S, Yajnik V (May 2016). "Inflammatory bowel disease and cancer: The role of inflammation, immunosuppression, and cancer treatment". World Journal of Gastroenterology (Review). 22 (20): 4794–4801. doi:10.3748/wjg.v22.i20.4794. PMC 4873872. PMID 27239106.
- ^ Stevens JP, Ballengee CR, Chandradevan R, Thompson AB, Schoen BT, Kugathasan S, et al. (October 2019). "Performance of Interferon-Gamma Release Assays for Tuberculosis Screening in Pediatric Inflammatory Bowel Disease". Journal of Pediatric Gastroenterology and Nutrition. 69 (4): e111–e116. doi:10.1097/MPG.0000000000002428. PMID 31261245. S2CID 195771593.
- ^ Lee CK, Wong SH, Lui G, Tang W, Tam LS, Ip M, et al. (July 2018). "A Prospective Study to Monitor for Tuberculosis During Anti-tumour Necrosis Factor Therapy in Patients With Inflammatory Bowel Disease and Immune-mediated Inflammatory Diseases". Journal of Crohn's & Colitis. 12 (8): 954–962. doi:10.1093/ecco-jcc/jjy057. PMID 29757355. S2CID 21673794.
- ^ Sandborn WJ, Vermeire S, Peyrin-Biroulet L, Dubinsky MC, Panes J, Yarur A, et al. (8 April 2023). "Etrasimod as induction and maintenance therapy for ulcerative colitis (ELEVATE): two randomised, double-blind, placebo-controlled, phase 3 studies". The Lancet. 401 (10383): 1159–1171. doi:10.1016/S0140-6736(23)00061-2. ISSN 0140-6736. PMID 36871574. S2CID 257286271.
- ^ Calkins BM (December 1989). "A meta-analysis of the role of smoking in inflammatory bowel disease". Digestive Diseases and Sciences. 34 (12): 1841–1854. doi:10.1007/BF01536701. PMID 2598752. S2CID 5775169.
- ^ Lakatos PL, Szamosi T, Lakatos L (December 2007). "Smoking in inflammatory bowel diseases: good, bad or ugly?". World Journal of Gastroenterology. 13 (46): 6134–6139. doi:10.3748/wjg.13.6134. PMC 4171221. PMID 18069751.
- ^ Calabrese E, Yanai H, Shuster D, Rubin DT, Hanauer SB (August 2012). "Low-dose smoking resumption in ex-smokers with refractory ulcerative colitis". Journal of Crohn's & Colitis. 6 (7): 756–762. doi:10.1016/j.crohns.2011.12.010. PMID 22398093.
- ^ Cosnes J (June 2004). "Tobacco and IBD: relevance in the understanding of disease mechanisms and clinical practice". Best Practice & Research. Clinical Gastroenterology. 18 (3): 481–496. doi:10.1016/j.bpg.2003.12.003. PMID 15157822.
- ^ Guslandi M (October 1999). "Nicotine treatment for ulcerative colitis". British Journal of Clinical Pharmacology. 48 (4): 481–484. doi:10.1046/j.1365-2125.1999.00039.x. PMC 2014383. PMID 10583016.
- ^ Sandborn WJ, Tremaine WJ, Offord KP, Lawson GM, Petersen BT, Batts KP, et al. (March 1997). "Transdermal nicotine for mildly to moderately active ulcerative colitis. A randomized, double-blind, placebo-controlled trial". Annals of Internal Medicine. 126 (5): 364–371. doi:10.7326/0003-4819-126-5-199703010-00004. PMID 9054280. S2CID 25745900.
- ^ Bonapace CR, Mays DA (1997). "The effect of mesalamine and nicotine in the treatment of inflammatory bowel disease". The Annals of Pharmacotherapy. 31 (7–8): 907–913. doi:10.1177/106002809703100719. PMID 9220055. S2CID 24122049.
- ^ Kennedy LD (September 1996). "Nicotine therapy for ulcerative colitis". The Annals of Pharmacotherapy. 30 (9): 1022–1023. PMID 8876866.
- ^ Rubin DT, Hanauer SB (August 2000). "Smoking and inflammatory bowel disease". European Journal of Gastroenterology & Hepatology. 12 (8): 855–862. doi:10.1097/00042737-200012080-00004. PMID 10958212.
- ^ a b Goddard AF, James MW, McIntyre AS, Scott BB, et al. (British Society of Gastroenterology) (October 2011). "Guidelines for the management of iron deficiency anaemia". Gut. 60 (10): 1309–1316. doi:10.1136/gut.2010.228874. PMID 21561874.
- ^ Gasche C, Berstad A, Befrits R, Beglinger C, Dignass A, Erichsen K, et al. (December 2007). "Guidelines on the diagnosis and management of iron deficiency and anemia in inflammatory bowel diseases". Inflammatory Bowel Diseases. 13 (12): 1545–1553. doi:10.1002/ibd.20285. PMID 17985376.
- ^ Mowat C, Cole A, Windsor A, Ahmad T, Arnott I, Driscoll R, et al. (May 2011). "Guidelines for the management of inflammatory bowel disease in adults". Gut. 60 (5): 571–607. doi:10.1136/gut.2010.224154. PMID 21464096. S2CID 8269837.
- ^ O'SULLIVAN PM (1964). "Drugs for Ulcerative Colitis". Canadian Medical Association Journal. 91 (21). Can Med Assoc J: 1123–1124. ISSN 0008-4409. PMC 1928363. PMID 14229762.
- ^ Camilleri M, Szarka L (14 November 2008). "Dysmotility of the Small Intestine and Colon". Textbook of Gastroenterology. Wiley. pp. 1108–1156. doi:10.1002/9781444303254.ch47. ISBN 978-1-4051-6911-0. S2CID 83791469.
- ^ Amanda Fernandez, OMS IV, Ronald Januchowski, DO, FACOFP (30 April 2020). "Osteopathic Primary Care Treatment Options for Ulcerative Colitis". Osteopathic Family Physician. 12 (3): 10–16. doi:10.33181/12031. ISSN 1877-573X. S2CID 219029672.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Sedano R, Quera R, Simian D, Yarur AJ (3 October 2019). "An approach to acute severe ulcerative colitis". Expert Review of Gastroenterology & Hepatology. 13 (10). Informa UK Limited: 943–955. doi:10.1080/17474124.2019.1681974. ISSN 1747-4124. PMID 31648574. S2CID 204891274.
- ^ Desai J, Elnaggar M, Hanfy AA, Doshi R (19 May 2020). "Toxic Megacolon: Background, Pathophysiology, Management Challenges and Solutions". Clinical and Experimental Gastroenterology. 13: 203–210. doi:10.2147/CEG.S200760. PMC 7245441. PMID 32547151.
- ^ Toruner, M.; Loftus, E.V.; Harmsen, W.S.; Zinsmeister, A.R.; Orenstein, R.; Sandborn, W.J.; Colombel, J.; Egan, L.J. Risk factors for opportunistic infections in patients with inflammatory bowel disease. Gastroenterology 2008, 134, 929–936
- ^ Farraye, F.A.; Melmed, G.Y.; Lichtenstein, G.R.; Kane, S.V. ACG Clinical Guideline: Preventive Care in Inflammatory Bowel Disease. Am. J. Gastroenterol. 2017, 112, 241–258.
- ^ Kucharzik, T.; Ellul, P.; Greuter, T.; Rahier, J.F.; Verstockt, B.; Abreu, C.; Albuquerque, A.; Allocca, M.; Esteve, M.; Farraye, F.A.; et al. ECCO Guidelines on the Prevention, Diagnosis, and Management of Infections in Inflammatory Bowel Disease. J. Crohn’s Colitis 2021, 15, 879–913.
- ^ Ananthakrishnan, A.N.; McGinley, E.L. Infection-related hospitalizations are associated with increased mortality in patients with inflammatory bowel diseases. J. Crohn’s Colitis 2013, 7, 107–112.
- ^ Winthrop, K.L.; Melmed, G.Y.; Vermeire, S.; Long, M.D.; Chan, G.; Pedersen, R.D.; Lawendy, N.; Thorpe, A.J.; Nduaka, C.I.; Su, C. Herpes Zoster Infection in Patients with Ulcerative Colitis Receiving Tofacitinib. Inflamm. Bowel Dis. 2018, 24, 2258–2265
- ^ Malhi, G.; Rumman, A.; Thanabalan, R.; Croitoru, K.; Silverberg, M.S.; Steinhart, A.H.; Nguyen, G.C. Vaccination in inflammatory bowel disease patients: Attitudes, knowledge, and uptake. J. Crohn’s Colitis 2015, 9, 439–444.
- ^ Costantino, A.; Michelon, M.; Noviello, D.; Macaluso, F.S.; Leone, S.; Bonaccorso, N.; Costantino, C.; Vecchi, M.; Caprioli, F., on behalf of AMICI Scientific Board. Attitudes towards Vaccinations in a National Italian Cohort of Patients with Inflammatory Bowel Disease. Vaccines 2023, 11, 1591.
- ^ a b Agabegi ED, Agabegi SS (2008). "Inflammatory bowel disease (IBD)". Step-Up to Medicine (Step-Up Series). Hagerstwon, MD: Lippincott Williams & Wilkins. pp. 152–156. ISBN 0-7817-7153-6.
- ^ Feller M, Huwiler K, Schoepfer A, Shang A, Furrer H, Egger M (February 2010). "Long-term antibiotic treatment for Crohn's disease: systematic review and meta-analysis of placebo-controlled trials". Clinical Infectious Diseases. 50 (4): 473–80. doi:10.1086/649923. PMID 20067425.
- ^ Prantera C, Scribano ML (July 2009). "Antibiotics and probiotics in inflammatory bowel disease: why, when, and how". Current Opinion in Gastroenterology. 25 (4): 329–33. doi:10.1097/MOG.0b013e32832b20bf. PMID 19444096.
- ^ "Living with a stoma". IBD Relief.
- ^ "Colectomy Not a Final Cure for Ulcerative Colitis, Data Show". www.mdedge.com. Retrieved 15 December 2019.
- ^ Pappou EP, Kiran RP (June 2016). "The Failed J Pouch". Clinics in Colon and Rectal Surgery. 29 (2): 123–129. doi:10.1055/s-0036-1580724. PMC 4882179. PMID 27247537.
- ^ a b Clarke WT, Feuerstein JD (August 2019). "Colorectal cancer surveillance in inflammatory bowel disease: Practice guidelines and recent developments". World Journal of Gastroenterology. 25 (30): 4148–4157. doi:10.3748/wjg.v25.i30.4148. PMC 6700690. PMID 31435169. S2CID 201114672.
- ^ Fedorak RN (November 2010). "Probiotics in the management of ulcerative colitis". Gastroenterology & Hepatology. 6 (11): 688–690. PMC 3033537. PMID 21437015.
- ^ a b c Kaur L, Gordon M, Baines PA, Iheozor-Ejiofor Z, Sinopoulou V, Akobeng AK (4 March 2020). Cochrane IBD Group (ed.). "Probiotics for induction of remission in ulcerative colitis". Cochrane Database of Systematic Reviews. 3 (3): CD005573. doi:10.1002/14651858.CD005573.pub3. PMC 7059959. PMID 32128795.
- ^ Iheozor-Ejiofor Z, Kaur L, Gordon M, Baines PA, Sinopoulou V, Akobeng AK (4 March 2020). Cochrane IBD Group (ed.). "Probiotics for maintenance of remission in ulcerative colitis". Cochrane Database of Systematic Reviews. 3 (3): CD007443. doi:10.1002/14651858.CD007443.pub3. PMC 7059960. PMID 32128794.
- ^ Borody TJ, Brandt LJ, Paramsothy S (January 2014). "Therapeutic faecal microbiota transplantation: current status and future developments". Current Opinion in Gastroenterology. 30 (1): 97–105. doi:10.1097/MOG.0000000000000027. PMC 3868025. PMID 24257037.
- ^ Narula N, Kassam Z, Yuan Y, Colombel JF, Ponsioen C, Reinisch W, et al. (October 2017). "Systematic Review and Meta-analysis: Fecal Microbiota Transplantation for Treatment of Active Ulcerative Colitis". Inflammatory Bowel Diseases. 23 (10): 1702–1709. doi:10.1097/MIB.0000000000001228. PMID 28906291.
- ^ Shi Y, Dong Y, Huang W, Zhu D, Mao H, Su P (2016). "Fecal Microbiota Transplantation for Ulcerative Colitis: A Systematic Review and Meta-Analysis". PLOS ONE. 11 (6): e0157259. Bibcode:2016PLoSO..1157259S. doi:10.1371/journal.pone.0157259. PMC 4905678. PMID 27295210.
- ^ Costello SP, Hughes PA, Waters O, Bryant RV, Vincent AD, Blatchford P, et al. (January 2019). "Effect of Fecal Microbiota Transplantation on 8-Week Remission in Patients With Ulcerative Colitis: A Randomized Clinical Trial". JAMA. 321 (2): 156–164. doi:10.1001/jama.2018.20046. PMC 6439766. PMID 30644982.
- ^ Hanai H, Iida T, Takeuchi K, Watanabe F, Maruyama Y, Andoh A, et al. (December 2006). "Curcumin maintenance therapy for ulcerative colitis: randomized, multicenter, double-blind, placebo-controlled trial". Clinical Gastroenterology and Hepatology. 4 (12): 1502–1506. doi:10.1016/j.cgh.2006.08.008. PMID 17101300.
- ^ a b Kumar S, Ahuja V, Sankar MJ, Kumar A, Moss AC (October 2012). "Curcumin for maintenance of remission in ulcerative colitis". The Cochrane Database of Systematic Reviews. 10: CD008424. doi:10.1002/14651858.CD008424.pub2. PMC 4001731. PMID 23076948.
- ^ Kafil TS, Nguyen TM, MacDonald JK, Chande N (November 2018). "Cannabis for the treatment of ulcerative colitis". The Cochrane Database of Systematic Reviews. 11 (11): CD012954. doi:10.1002/14651858.CD012954.pub2. PMC 6516819. PMID 30406638.
- ^ Sinopoulou V, Gordon M, Dovey TM, Akobeng AK, et al. (Cochrane Gut Group) (July 2021). "Interventions for the management of abdominal pain in ulcerative colitis". The Cochrane Database of Systematic Reviews. 2021 (7): CD013589. doi:10.1002/14651858.CD013589.pub2. PMC 8407332. PMID 34291816.
- ^ De Souza, H.; Fiocchi, C.; Iliopoulos, D. The IBD interactome: An integrated view of aetiology, pathogenesis and therapy. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 739–749
- ^ Roncoroni L, Gori R, Elli L, Tontini GE, Doneda L, Norsa L, et al. (10 February 2022). "Nutrition in Patients with Inflammatory Bowel Diseases: A Narrative Review". Nutrients. 14 (4): 751. doi:10.3390/nu14040751. ISSN 2072-6643. PMC 8879392. PMID 35215401.
 This article incorporates text from this source, which is available under the CC BY 4.0 license.
This article incorporates text from this source, which is available under the CC BY 4.0 license.
- ^ Fracas E, Costantino A, Vecchi M, Buoli M. Depressive and Anxiety Disorders in Patients with Inflammatory Bowel Diseases: Are There Any Gender Differences? International Journal of Environmental Research and Public Health. 2023; 20(13):6255. https://doi.org/10.3390/ijerph20136255
- ^ Barberio B, Zamani M, Black CJ, Savarino EV, Ford AC. Prevalence of symptoms of anxiety and depression in patients with inflammatory bowel disease: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2021 May;6(5):359-370. doi: 10.1016/S2468-1253(21)00014-5
- ^ Seaton N, Hudson J, Harding S, Norton S, Mondelli V, Jones AS, et al. (2 February 2024). "Do interventions for mood improve inflammatory biomarkers in inflammatory bowel disease?: a systematic review and meta-analysis". eBioMedicine. 100: 104910. doi:10.1016/j.ebiom.2023.104910. ISSN 2352-3964. PMC 10878994. PMID 38272759.
- ^ "Improving mood reduces inflammation in inflammatory bowel disease". NIHR Evidence. 17 July 2024. doi:10.3310/nihrevidence_63192.
- ^ Kevans D, Murthy S, Mould DR, Silverberg MS (May 2018). "Accelerated Clearance of Infliximab is Associated With Treatment Failure in Patients With Corticosteroid-Refractory Acute Ulcerative Colitis". Journal of Crohn's & Colitis. 12 (6): 662–669. doi:10.1093/ecco-jcc/jjy028. PMID 29659758.
- ^ Horio Y, Uchino M, Bando T, Chohno T, Sasaki H, Hirata A, et al. (May 2017). "Rectal-sparing type of ulcerative colitis predicts lack of response to pharmacotherapies". BMC Surgery. 17 (1): 59. doi:10.1186/s12893-017-0255-5. PMC 5437574. PMID 28526076.
- ^ Osterman MT, Lichtenstein GR (2010). "Ulcerative Colitis". Sleisenger and Fordtran's Gastrointestinal and Liver Disease. pp. 1975–2013.e9. doi:10.1016/B978-1-4160-6189-2.00112-8. ISBN 9781416061892.
- ^ Patil DT, Odze RD (August 2017). "Backwash Is Hogwash: The Clinical Significance of Ileitis in Ulcerative Colitis". The American Journal of Gastroenterology. 112 (8): 1211–1214. doi:10.1038/ajg.2017.182. PMID 28631729. S2CID 10801391.
- ^ Leighton JA, Shen B, Baron TH, Adler DG, Davila R, Egan JV, et al. (April 2006). "ASGE guideline: endoscopy in the diagnosis and treatment of inflammatory bowel disease". Gastrointestinal Endoscopy. 63 (4): 558–565. doi:10.1016/j.gie.2006.02.005. PMID 16564852.
- ^ a b Jess T, Gamborg M, Munkholm P, Sørensen TI (March 2007). "Overall and cause-specific mortality in ulcerative colitis: meta-analysis of population-based inception cohort studies". The American Journal of Gastroenterology. 102 (3): 609–617. doi:10.1111/j.1572-0241.2006.01000.x. PMID 17156150. S2CID 2086542.
- ^ da Silva BC, Lyra AC, Rocha R, Santana GO (July 2014). "Epidemiology, demographic characteristics and prognostic predictors of ulcerative colitis". World Journal of Gastroenterology. 20 (28): 9458–9467. doi:10.3748/wjg.v20.i28.9458. PMC 4110577. PMID 25071340.
- ^ Nguyen M, Bradford K, Zhang X, Shih DQ (January 2011). "Cytomegalovirus Reactivation in Ulcerative Colitis Patients". Ulcers. 2011: 1–7. doi:10.1155/2011/282507. PMC 3124815. PMID 21731826.
- ^ Vos T, Allen C, Arora M, Barber RM, Bhutta ZA, Brown A, et al. (GBD 2015 Disease and Injury Incidence and Prevalence Collaborators) (October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1545–1602. doi:10.1016/S0140-6736(16)31678-6. PMC 5055577. PMID 27733282.
- ^ a b Adams JG (2012). Emergency Medicine E-Book: Clinical Essentials (Expert Consult – Online). Elsevier Health Sciences. p. 304. ISBN 978-1455733941.
- ^ Karlinger K, Györke T, Makö E, Mester A, Tarján Z (September 2000). "The epidemiology and the pathogenesis of inflammatory bowel disease". European Journal of Radiology. 35 (3): 154–167. doi:10.1016/s0720-048x(00)00238-2. PMID 11000558.
- ^ Podolsky DK (August 2002). "Inflammatory bowel disease". The New England Journal of Medicine. 347 (6): 417–429. doi:10.1056/NEJMra020831. PMID 12167685.
- ^ Schmidt JA, Marshall J, Hayman MJ (December 1985). "Identification and characterization of the chicken transferrin receptor". The Biochemical Journal. 232 (3): 735–741. doi:10.1042/bj2320735. PMC 1152945. PMID 3004417.
- ^ Shivananda S, Lennard-Jones J, Logan R, Fear N, Price A, Carpenter L, et al. (November 1996). "Incidence of inflammatory bowel disease across Europe: is there a difference between north and south? Results of the European Collaborative Study on Inflammatory Bowel Disease (EC-IBD)". Gut. 39 (5): 690–697. doi:10.1136/gut.39.5.690. PMC 1383393. PMID 9014768.
- ^ Sonnenberg A, McCarty DJ, Jacobsen SJ (January 1991). "Geographic variation of inflammatory bowel disease within the United States". Gastroenterology. 100 (1): 143–149. doi:10.1016/0016-5085(91)90594-B. PMID 1983816.
- ^ Burisch J, Pedersen N, Čuković-Čavka S, Brinar M, Kaimakliotis I, Duricova D, et al. (April 2014). "East-West gradient in the incidence of inflammatory bowel disease in Europe: the ECCO-EpiCom inception cohort". Gut. 63 (4): 588–597. doi:10.1136/gutjnl-2013-304636. hdl:2336/325171. PMID 23604131. S2CID 25069828.
- ^ Andersson RE, Olaison G, Tysk C, Ekbom A (March 2001). "Appendectomy and protection against ulcerative colitis". The New England Journal of Medicine. 344 (11): 808–814. doi:10.1056/NEJM200103153441104. PMID 11248156.
- ^ a b Boyko EJ, Koepsell TD, Perera DR, Inui TS (March 1987). "Risk of ulcerative colitis among former and current cigarette smokers". The New England Journal of Medicine. 316 (12): 707–710. doi:10.1056/NEJM198703193161202. PMID 3821808.
- ^ a b "Epidemiology of the IBD". Centers for Disease Control and Prevention (CDC). Archived from the original on 23 February 2017. Retrieved 23 February 2017.
- ^ Makhlouf GM, Zfass AM, Said SI, Schebalin M (April 1978). "Effects of synthetic vasoactive intestinal peptide (VIP), secretin and their partial sequences on gastric secretion". Proceedings of the Society for Experimental Biology and Medicine. 157 (4): 565–568. doi:10.3181/00379727-157-40097. PMID 349569. S2CID 40543366.
- ^ "Ulcerative colitis: management". National Institute for Health and Care Excellence. 3 May 2019.
- ^ a b Mulder D, Noble A, Justinich C, Duffin J (1 May 2014). "A tale of two diseases: The history of inflammatory bowel disease". Journal of Crohn's and Colitis. 8 (5): 341–348. doi:10.1016/j.crohns.2013.09.009. PMID 24094598.
- ^ a b Summers RW, Elliott DE, Urban JF, Thompson RA, Weinstock JV (April 2005). "Trichuris suis therapy for active ulcerative colitis: a randomized controlled trial". Gastroenterology. 128 (4): 825–832. doi:10.1053/j.gastro.2005.01.005. PMID 15825065.
- ^ a b Bennett CF, Condon TC, Grimm S, Chan H, Chiang MY (1994). "Inhibition of endothelial cell-leukocyte adhesion molecule expression with antisense oligonucleotides". The Journal of Immunology. 152 (1): 3530–40. doi:10.4049/jimmunol.152.7.3530. S2CID 34563008.
- ^ Jones SC, Banks RE, Haidar A, Gearing AJ, Hemingway IK, Ibbotson SH, et al. (May 1995). "Adhesion molecules in inflammatory bowel disease". Gut. 36 (5): 724–730. doi:10.1136/gut.36.5.724. PMC 1382677. PMID 7541009.
- ^ van Deventer SJ, Wedel MK, Baker BF, Xia S, Chuang E, Miner PB (May 2006). "A phase II dose ranging, double-blind, placebo-controlled study of alicaforsen enema in subjects with acute exacerbation of mild to moderate left-sided ulcerative colitis". Alimentary Pharmacology & Therapeutics. 23 (10): 1415–1425. doi:10.1111/j.1365-2036.2006.02910.x. PMID 16669956. S2CID 31495688.
- ^ Ghouri YA, Richards DM, Rahimi EF, Krill JT, Jelinek KA, DuPont AW (9 December 2014). "Systematic review of randomized controlled trials of probiotics, prebiotics, and synbiotics in inflammatory bowel disease". Clinical and Experimental Gastroenterology. 7: 473–487. doi:10.2147/CEG.S27530. PMC 4266241. PMID 25525379.
- ^ a b Strøbæk D, Brown DT, Jenkins DP, Chen YJ, Coleman N, Ando Y, et al. (January 2013). "NS6180, a new K(Ca) 3.1 channel inhibitor prevents T-cell activation and inflammation in a rat model of inflammatory bowel disease". British Journal of Pharmacology. 168 (2): 432–444. doi:10.1111/j.1476-5381.2012.02143.x. PMC 3572569. PMID 22891655.
- ^ a b Bennike TB, Carlsen TG, Ellingsen T, Bonderup OK, Glerup H, Bøgsted M, et al. (September 2015). "Neutrophil Extracellular Traps in Ulcerative Colitis: A Proteome Analysis of Intestinal Biopsies". Inflammatory Bowel Diseases. 21 (9): 2052–2067. doi:10.1097/MIB.0000000000000460. PMC 4603666. PMID 25993694.
- ^ Kirov S, Sasson A, Zhang C, Chasalow S, Dongre A, Steen H, et al. (February 2019). "Degradation of the extracellular matrix is part of the pathology of ulcerative colitis". Molecular Omics. 15 (1): 67–76. doi:10.1039/C8MO00239H. PMID 30702115.
- ^ Raithel M, Winterkamp S, Weidenhiller M, Müller S, Hahn EG (July 2007). "Combination therapy using fexofenadine, disodium cromoglycate, and a hypoallergenic amino acid-based formula induced remission in a patient with steroid-dependent, chronically active ulcerative colitis". International Journal of Colorectal Disease. 22 (7): 833–839. doi:10.1007/s00384-006-0120-y. PMID 16944185. S2CID 2605447.
- ^ a b Dhaneshwar S, Gautam H (August 2012). "Exploring novel colon-targeting antihistaminic prodrug for colitis" (PDF). Journal of Physiology and Pharmacology. 63 (4): 327–337. PMID 23070081.
- ^ Vermeire S, O'Byrne S, Keir M, Williams M, Lu TT, Mansfield JC, et al. (July 2014). "Etrolizumab as induction therapy for ulcerative colitis: a randomised, controlled, phase 2 trial". Lancet. 384 (9940): 309–318. doi:10.1016/S0140-6736(14)60661-9. PMID 24814090. S2CID 7369482.
- ^ a b Rosenfeld G, Parker CE, MacDonald JK, Bressler B (December 2015). "Etrolizumab for induction of remission in ulcerative colitis". The Cochrane Database of Systematic Reviews. 2015 (12): CD011661. doi:10.1002/14651858.CD011661.pub2. PMC 8612697. PMID 26630451.
- ^ Makker J, Hommes DW (2016). "Etrolizumab for ulcerative colitis: the new kid on the block?". Expert Opinion on Biological Therapy. 16 (4): 567–572. doi:10.1517/14712598.2016.1158807. PMID 26914639. S2CID 24706213.
- ^ Taylor N (3 February 2022). "Roche lets go of etro, dumping phase 3 Crohn's prospect 18 months after posting weak colitis data". Fierce Biotech. Retrieved 12 December 2023.
- ^ Abreu MT, Plevy S, Sands BE, Weinstein R (2007). "Selective leukocyte apheresis for the treatment of inflammatory bowel disease". Journal of Clinical Gastroenterology. 41 (10): 874–888. doi:10.1097/MCG.0b013e3180479435. PMID 18090155. S2CID 36724094.
- ^ Vernia P, D'Ovidio V, Meo D (October 2010). "Leukocytapheresis in the treatment of inflammatory bowel disease: Current position and perspectives". Transfusion and Apheresis Science. 43 (2): 227–229. doi:10.1016/j.transci.2010.07.023. PMID 20817610.
Further reading
[edit]- Rubin DT, Ananthakrishnan AN, Siegel CA, Sauer BG, Long MD (March 2019). "ACG Clinical Guideline: Ulcerative Colitis in Adults". The American Journal of Gastroenterology. 114 (3): 384–413. doi:10.14309/ajg.0000000000000152. PMID 30840605. S2CID 73473272.

- Torpy JM, Lynm C, Golub RM (January 2012). "JAMA patient page. Ulcerative colitis". JAMA. 307 (1): 104. doi:10.1001/jama.2011.1889. PMID 22215172.
External links
[edit]- MedlinePlus ulcerative colitis page
 Media related to Ulcerative colitis at Wikimedia Commons
Media related to Ulcerative colitis at Wikimedia Commons
