Guanidine
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Guanidine[1] | |||
| Other names
Iminomethanediamine
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 506044 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.003.656 | ||
| EC Number |
| ||
| 100679 | |||
| MeSH | Guanidine | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| CH5N3 | |||
| Molar mass | 59.072 g·mol−1 | ||
| Melting point | 50 °C (122 °F; 323 K) | ||
| log P | −1.251 | ||
| Basicity (pKb) | 0.4[2] | ||
| Conjugate acid | Guanidinium | ||
| Thermochemistry | |||
Std enthalpy of
formation (ΔfH⦵298) |
−57 – −55 kJ mol−1 | ||
Std enthalpy of
combustion (ΔcH⦵298) |
−1.0511 – −1.0531 MJ mol−1 | ||
| Pharmacology | |||
| Pharmacokinetics: | |||
| 7–8 hours | |||
| Hazards | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
475 mg/kg (oral, rat)[3] | ||
| Related compounds | |||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
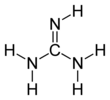
Guanidine is the compound with the formula HNC(NH2)2. It is a colourless solid that dissolves in polar solvents. It is a strong base that is used in the production of plastics and explosives. It is found in urine predominantly in patients experiencing renal failure.[4] A guanidine moiety also appears in larger organic molecules, including on the side chain of arginine.
Structure
[edit]Guanidine can be thought of as a nitrogenous analogue of carbonic acid. That is, the C=O group in carbonic acid is replaced by a C=NH group, and each OH is replaced by a NH
2 group.[5] Isobutene can be seen as the carbon analogue in much the same way. A detailed crystallographic analysis of guanidine was elucidated 148 years after its first synthesis, despite the simplicity of the molecule.[6] In 2013, the positions of the hydrogen atoms and their displacement parameters were accurately determined using single-crystal neutron diffraction.[7]
Production
[edit]Guanidine can be obtained from natural sources, being first isolated in 1861 by Adolph Strecker via the oxidative degradation of an aromatic natural product, guanine, isolated from Peruvian guano.[8][9]
A laboratory method of producing guanidine is gentle (180-190 °C) thermal decomposition of dry ammonium thiocyanate in anhydrous conditions:
- 3 NH4SCN → 2 CH5N3 + H2S + CS2
The commercial route involves a two step process starting with the reaction of dicyandiamide with ammonium salts. Via the intermediacy of biguanidine, this ammonolysis step affords salts of the guanidinium cation (see below). In the second step, the salt is treated with base, such as sodium methoxide.[8]
Isothiouronium salts (S-alkylated thioureas) react with amines to give guanidinium salts:[10]
- RNH2 + [CH3SC(NH2)2]+X− → [RN(H)C(NH2)2]+X− + CH3SH
The resulting guanidinium ions can often be deprotonated to give the guanidine. This approach is sometimes called the Rathke synthesis, in honor of its discoverer. after Bernhard Rathke[11][12]
Chemistry
[edit]Guanidinium cation
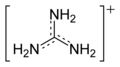
[edit]The conjugate acid is called the guanidinium cation, (C(NH
2)+
3). This planar, symmetric ion consists of three amino groups each bonded to the central carbon atom with a covalent bond of order 4/3. It is a highly stable +1 cation in aqueous solution due to the efficient resonance stabilization of the charge and efficient solvation by water molecules. As a result, its pKaH is 13.6[2] (pKb of 0.4) meaning that guanidine is a very strong base in water; in neutral water, it exists almost exclusively as guanidinium. Due to this, most guanidine derivatives are salts containing the conjugate acid.
-
canonical forms
Testing for guanidine
[edit]Guanidine can be selectively detected using sodium 1,2-naphthoquinone-4-sulfonic acid (Folin's reagent) and acidified urea.[13]
Uses
[edit]Industry
[edit]The main salt of commercial interest is the nitrate [C(NH
2)3]NO
3. It is used as a propellant, for example in air bags.
Medicine
[edit]Since the Middle Ages in Europe, guanidine has been used to treat diabetes as the active antihyperglycemic ingredient in French lilac. Due to its long-term hepatotoxicity, further research for blood sugar control was suspended at first after the discovery of insulin. Later development of nontoxic, safe biguanides led to the long-used first-line diabetes control medicine metformin, introduced to Europe in the 1950s & United States in 1995 and now prescribed to over 17 million patients per year in the US.[14][15]
Guanidinium chloride[14] is a now-controversial adjuvant in treatment of botulism. Recent studies have shown some significant subsets of patients who see no improvement after the administration of this drug.[16]
Biochemistry
[edit]Guanidine exists protonated, as guanidinium, in solution at physiological pH.
Guanidinium chloride (also known as guanidine hydrochloride) has chaotropic properties and is used to denature proteins. Guanidinium chloride is known to denature proteins with a linear relationship between concentration and free energy of unfolding. In aqueous solutions containing 6 M guanidinium chloride, almost all proteins lose their entire secondary structure and become randomly coiled peptide chains. Guanidinium thiocyanate is also used for its denaturing effect on various biological samples.
Recent studies suggest that guanidinium is produced by bacteria as a toxic byproduct. To alleviate the toxicity of guanidinium, bacteria have developed a class of transporters known as guanidinium exporters or Gdx proteins to expel the extra amounts of this ion to the outside of the cell.[17] Gdx proteins, are highly selective for guanidinium and mono-substituted guanidinyl compounds and share an overlapping set of non-canonical substrates with drug exporter EmrE.[18]
Other
[edit]Guanidinium hydroxide is the active ingredient in some non-lye hair relaxers.
Guanidine derivatives
[edit]
Guanidines are a group of organic compounds sharing a common functional group with the general structure (R
1R
2N)(R
3R
4N)C=N−R
5. The central bond within this group is that of an imine, and the group is related structurally to amidines and ureas. Examples of guanidines are arginine, triazabicyclodecene, saxitoxin, and creatine.
Galegine is an isoamylene guanidine.[19]
See also
[edit]References
[edit]- ^ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. p. 883. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ^ a b Perrin DD (1972). Dissociation Constants of Organic Bases in Aqueous Solution (Supplement ed.). London: Butterworths.
- ^ "Guanidine hydrochloride". ChemIDplus. National Library of Medicine. Archived from the original on 2014-08-12. Retrieved 2014-08-10.
- ^ Sawynok J, Dawborn JK (1975). "Plasma concentration and urinary excretion of guanidine derivatives in normal subjects and patients with renal failure". Clinical and Experimental Pharmacology & Physiology. 2 (1): 1–15. doi:10.1111/j.1440-1681.1975.tb02368.x. PMID 1126056. S2CID 41794868.
- ^ Göbel M, Klapötke TM (August 2007). "First structural characterization of guanidine, HN=C(NH(2))(2)". Chemical Communications. 43 (30): 3180–3182. doi:10.1039/B705100J. PMID 17653381.
- ^ Yamada T, Liu X, Englert U, Yamane H, Dronskowski R (June 2009). "Solid-state structure of free base guanidine achieved at last". Chemistry: A European Journal. 15 (23): 5651–5655. doi:10.1002/chem.200900508. PMID 19388036.
- ^ Sawinski PK, Meven M, Englert U, Dronskowski R (2013). "Single-Crystal Neutron Diffraction Study on Guanidine, CN3H5". Crystal Growth & Design. 13 (4): 1730–5. doi:10.1021/cg400054k.
- ^ a b Güthner T, Mertschenk B, Schulz B. "Guanidine and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a12_545.pub2. ISBN 978-3527306732.
- ^ Strecker A (1861). "Untersuchungen über die chemischen Beziehungen zwischen Guanin, Xanthin, Theobromin, Caffeïn und Kreatinin" [Studies on the chemical relationships between guanine, xanthine, theobromine, caffeine and creatinine]. Liebigs Ann. Chem. 118 (2): 151–177. doi:10.1002/jlac.18611180203. Archived from the original on 2021-07-16. Retrieved 2019-07-02.
- ^ Palmer, David C. (2001). "S-Methylisothiourea". E-EROS Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rm199s. ISBN 0471936235.
- ^ "Heinrich Bernhard Rathke. (1840-1923)". Berichte der Deutschen Chemischen Gesellschaft (A and B Series). 57 (9): A83 – A92. 8 October 1924. doi:10.1002/cber.19240570929.
- ^ Rathke, B. (July 1881). "Ueber Derivate und Constitution des Schwefelharnstoffs". Berichte der Deutschen Chemischen Gesellschaft. 14 (2): 1774–1780. doi:10.1002/cber.18810140247.
- ^ Sullivan MX (1935-10-01). "A Colorimetric Test for Guanidine". Proceedings of the Society for Experimental Biology and Medicine. 33 (1): 106–108. doi:10.3181/00379727-33-8270C. ISSN 0037-9727. S2CID 88290359.
- ^ a b Blaslov K, Naranđa FS, Kruljac I, Renar IP (December 2018). "Treatment approach to type 2 diabetes: Past, present and future". World Journal of Diabetes. 9 (12): 209–219. doi:10.4239/wjd.v9.i12.209. PMC 6304295. PMID 30588282.
- ^ "The Top 300 of 2019". clincalc.com. Archived from the original on 2021-02-12. Retrieved 2022-02-17.
- ^ Brook I (2001). Pediatric Anaerobic Infections: Diagnosis and Management (3rd ed.). Taylor & Francis. p. 529. ISBN 0824741862.
- ^ Kermani AA, Macdonald CB, Gundepudi R, Stockbridge RB (March 2018). "Guanidinium export is the primal function of SMR family transporters". Proceedings of the National Academy of Sciences of the United States of America. 115 (12): 3060–3065. Bibcode:2018PNAS..115.3060K. doi:10.1073/pnas.1719187115. PMC 5866581. PMID 29507227.
- ^ Kermani AA, Macdonald CB, Burata OE, Ben Koff B, Koide A, Denbaum E, et al. (November 2020). "The structural basis of promiscuity in small multidrug resistance transporters". Nature Communications. 11 (1): 6064. Bibcode:2020NatCo..11.6064K. doi:10.1038/s41467-020-19820-8. PMC 7695847. PMID 33247110.
- ^ Witters LA (October 2001). "The blooming of the French lilac". The Journal of Clinical Investigation. 108 (8): 1105–1107. doi:10.1172/JCI14178. PMC 209536. PMID 11602616.