Feldspar
| Feldspar | |
|---|---|
 Feldspar minerals. Clockwise from top left: Orthoclase, Albite, Microcline, and an indeterminate plagioclase | |
| General | |
| Category | Tectosilicate |
| Formula (repeating unit) | KAlSi 3O 8 – NaAlSi 3O 8 – CaAl 2Si 2O 8 |
| IMA symbol | Fsp[1] |
| Crystal system | Triclinic or monoclinic |
| Identification | |
| Color | pink, white, gray, brown, blue |
| Cleavage | two or three |
| Fracture | along cleavage planes |
| Mohs scale hardness | 6.0–6.5 |
| Luster | Vitreous |
| Streak | white |
| Diaphaneity | opaque |
| Specific gravity | 2.55–2.76 |
| Density | 2.56 |
| Refractive index | 1.518–1.526 |
| Birefringence | first order |
| Pleochroism | none |
| Other characteristics | exsolution lamellae common |
| References | [2] |
Feldspar (/ˈfɛl(d)ˌspɑːr/ FEL(D)-spar; sometimes spelled felspar) is a group of rock-forming aluminium tectosilicate minerals, also containing other cations such as sodium, calcium, potassium, or barium.[3] The most common members of the feldspar group are the plagioclase (sodium-calcium) feldspars and the alkali (potassium-sodium) feldspars.[4] Feldspars make up about 60% of the Earth's crust[3] and 41% of the Earth's continental crust by weight.[5][6]
Feldspars crystallize from magma as both intrusive and extrusive igneous rocks[7] and are also present in many types of metamorphic rock.[8] Rock formed almost entirely of calcic plagioclase feldspar is known as anorthosite.[9] Feldspars are also found in many types of sedimentary rocks.[10]
Etymology
[edit]The name feldspar derives from the German Feldspat, a compound of the words Feld ("field") and Spat ("flake"). Spat had long been used as the word for "a rock easily cleaved into flakes"; Feldspat was introduced in the 18th century as a more specific term, referring perhaps to its common occurrence in rocks found in fields (Urban Brückmann, 1783) or to its occurrence as "fields" within granite and other minerals (René-Just Haüy, 1804).[11] The change from Spat to -spar was influenced by the English word spar,[12] meaning a non-opaque mineral with good cleavage.[13] Feldspathic refers to materials that contain feldspar. The alternate spelling, felspar, has fallen out of use. The term 'felsic', meaning light coloured minerals such as quartz and feldspars, is an acronymic word derived from feldspar and silica, unrelated to the obsolete spelling 'felspar'.
Compositions
[edit]
The feldspar group of minerals consists of tectosilicates, silicate minerals in which silicon ions are linked by shared oxygen ions to form a three-dimensional network. Compositions of major elements in common feldspars can be expressed in terms of three endmembers:
- potassium feldspar (K-spar) endmember KAlSi3O8[14]
- albite endmember NaAlSi3O8[14]
- anorthite endmember CaAl2Si2O8[14]
Solid solutions between K-feldspar and albite are called alkali feldspar.[14] Solid solutions between albite and anorthite are called plagioclase,[14] or, more properly, plagioclase feldspar. Only limited solid solution occurs between K-feldspar and anorthite, and in the two other solid solutions, immiscibility occurs at temperatures common in the crust of the Earth. Albite is considered both a plagioclase and alkali feldspar.
The ratio of alkali feldspar to plagioclase feldspar, together with the proportion of quartz, is the basis for the QAPF classification of igneous rock.[15][16][17] Calcium-rich plagioclase is the first feldspar to crystallize from cooling magma, then the plagioclase becomes increasingly sodium-rich as crystallization continues. This defines the continuous Bowen's reaction series. K-feldspar is the final feldspar to crystallize from the magma.[18][19]
Alkali feldspars
[edit]Alkali feldspars are grouped into two types: those containing potassium in combination with sodium, aluminium, or silicon; and those where potassium is replaced by barium. The first of these include:
- orthoclase (monoclinic)[20] KAlSi3O8
- sanidine (monoclinic)[21] (K,Na)AlSi3O8
- microcline (triclinic)[22] KAlSi3O8
- anorthoclase (triclinic) (Na,K)AlSi3O8
Potassium and sodium feldspars are not perfectly miscible in the melt at low temperatures, therefore intermediate compositions of the alkali feldspars occur only in higher temperature environments.[23] Sanidine is stable at the highest temperatures, and microcline at the lowest.[20][21] Perthite is a typical texture in alkali feldspar, due to exsolution of contrasting alkali feldspar compositions during cooling of an intermediate composition. The perthitic textures in the alkali feldspars of many granites can be seen with the naked eye.[24] Microperthitic textures in crystals are visible using a light microscope, whereas cryptoperthitic textures can be seen only with an electron microscope.
Ammonium feldspar
[edit]Buddingtonite is an ammonium feldspar with the chemical formula: NH4AlSi3O8.[25] It is a mineral associated with hydrothermal alteration of the primary feldspar minerals.
Barium feldspars
[edit]Barium feldspars form as the result of the substitution of barium for potassium in the mineral structure. Barium feldspars are sometimes classified as a separate group of feldspars,[4] and sometimes they are classified as a sub-group of alkali feldspars.[26]
The barium feldspars are monoclinic and include the following:
- celsian BaAl2Si2O8[27]
- hyalophane (K,Ba)(Al,Si)4O8[28]
Plagioclase feldspars
[edit]The plagioclase feldspars are triclinic. The plagioclase series follows (with percent anorthite in parentheses):
- albite (0 to 10) NaAlSi3O8
- oligoclase (10 to 30) (Na,Ca)(Al,Si)AlSi2O8
- andesine (30 to 50) NaAlSi
3O
8 – CaAl
2Si
2O
8 - labradorite (50 to 70) (Ca,Na)Al(Al,Si)Si2O8
- bytownite (70 to 90) (NaSi,CaAl)AlSi2O8
- anorthite (90 to 100) CaAl2Si2O8
Intermediate compositions of exsolve to two feldspars of contrasting composition during cooling, but diffusion is much slower than in alkali feldspar, and the resulting two-feldspar intergrowths typically are too fine-grained to be visible with optical microscopes. The immiscibility gaps in the plagioclase solid solutions are complex compared to the gap in the alkali feldspars. The play of colours visible in some feldspar of labradorite composition is due to very fine-grained exsolution lamellae known as Bøggild intergrowth. The specific gravity in the plagioclase series increases from albite (2.62) to anorthite (2.72–2.75).
Structure
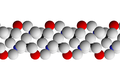
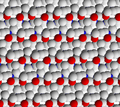
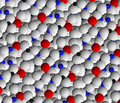
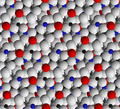
[edit]The structure of a feldspar crystal is based on aluminosilicate tetrahedra. Each tetrahedron consists of an aluminium or silicon ion surrounded by four oxygen ions. Each oxygen ion, in turn, is shared by a neighbouring tetrahedron to form a three-dimensional network. The structure can be visualized as long chains of aluminosilicate tetrahedra, sometimes described as crankshaft chains because their shape is kinked. Each crankshaft chain links to neighbouring crankshaft chains to form a three-dimensional network of fused four-member rings. The structure is open enough for cations (typically sodium, potassium, or calcium) to fit into the structure and provide charge balance.[29]
-
Diagram showing part of a crankshaft chain of feldspar
-
Feldspar crystal structure viewed along the c axis
-
Feldspar crystal structure viewed along the a axis
-
Feldspar crystal structure viewed along the b axis
Weathering
[edit]Chemical weathering of feldspars happens by hydrolysis and produces clay minerals, including illite, smectite, and kaolinite. Hydrolysis of feldspars begins with the feldspar dissolving in water, which happens best in acidic or basic solutions and less well in neutral ones.[30] The speed at which feldspars are weathered is controlled by how quickly they are dissolved.[30] Dissolved feldspar reacts with H+ or OH− ions and precipitates clays. The reaction also produces new ions in solution, with the variety of ions controlled by the type of feldspar reacting.
The abundance of feldspars in the Earth's crust means that clays are very abundant weathering products.[31] About 40% of minerals in sedimentary rocks are clays and clays are the dominant minerals in the most common sedimentary rocks, mudrocks.[32] They are also an important component of soils.[32] Feldspar that has been replaced by clay looks chalky compared to more crystalline and glassy unweathered feldspar grains.[33]
Feldspars, especially plagioclase feldspars, are not very stable at the Earth's surface due to their high formation temperature.[32] This lack of stability is why feldspars are easily weathered to clays. Because of this tendency to weather easily, feldspars are usually not prevalent in sedimentary rocks. Sedimentary rocks that contain large amounts of feldspar indicate that the sediment did not undergo much chemical weathering before being buried. This means it was probably transported a short distance in cold and/or dry conditions that did not promote weathering, and that it was quickly buried by other sediment.[34] Sandstones with large amounts of feldspar are called arkoses.[34]
Applications
[edit]Feldspar is a common raw material used in glassmaking, ceramics, and to some extent as filler and an extender in paint, plastics, and rubber. In the US, about 66% of feldspar is consumed in glassmaking, including glass containers and glass fibre. Ceramics (including electrical insulators, sanitaryware, tableware and tile) and other uses, such as fillers, accounted for the remainder.[35]
Glass: Feldspar provides both K2O and Na2O for fluxing, and Al2O3 and CaO as stabilizers. As an important source of Al2O3 for glassmaking, feldspar is valued for its low iron and refractory mineral content, a low cost per unit of Al2O3, no volatiles and no waste.[36]
Ceramics: Feldspars are used in the ceramic industry as a flux to form a glassy phase in bodies during firing, and thus promote vitrification. They also are used as a source of alkalies and alumina in glazes.[36] The composition of feldspar used in different ceramic formulations varies depending on various factors, including the properties of the individual grade, the other raw materials and the requirements of the finished products. However, typical additions include: tableware, 15% to 30% feldspar; high-tension electrical porcelains, 25% to 35%; sanitaryware, 25%; wall tile, 0% to 10%; and dental porcelain up to 80% feldspar.[37]
Earth sciences: In earth sciences and archaeology, feldspars are used for potassium-argon dating, argon-argon dating and luminescence dating.
Minor use: Some household cleaners (such as Bar Keepers Friend and Bon Ami) use feldspar to give a mild abrasive action.[38]
Production
[edit]The USGS estimated global production of feldspar in 2020 to be 26 million tonnes, with the top four producing countries being: China 2 million tonnes; India 5 million tonnes; Italy 4 million; Turkey 7.6 million tonnes.[39]
Commercial grades
[edit]Typical mineralogical and chemical analyses of three commercial grades used in ceramics are:[40]
| Product name | Norfloat K | Forshammar | FFF K6 |
|---|---|---|---|
| Country | Norway | Sweden | Finland |
| Producing company | North Cape | Sibelco | Sibelco |
| Albite, % | 23 | 40 | 41 |
| Microcline, % | 71 | 23 | 37 |
| Anorthite, % | 3 | - | 4 |
| Quartz , % | 3 | 33 | 8 |
| SiO2, % | 65.9 | 75.7 | 67.9 |
| Al2O3, % | 18.6 | 14.1 | 18.3 |
| Fe2O3, % | 0.07 | 0.15 | 0.11 |
| TiO2, % | - | 0.02 | 0.01 |
| CaO, % | 0.40 | 0.30 | 0.70 |
| MgO, % | - | 0.10 | 0.01 |
| K2O, % | 11.8 | 3.8 | 6.4 |
| Na2O, % | 2.9 | 5.0 | 5.5 |
| LOI, % | 0.2 | 0.5 | 0.2 |
Extraterrestrial
[edit]In October 2012, the Curiosity rover found high feldspar content in a Mars rock.[41]
Gallery
[edit]-
Specimen of rare plumbian (lead-rich) feldspar
-
Crystallized white feldspar, with an upright 4 cm aquamarine crystal perched on it
-
Feldspar and moonstone, from Sonora, Mexico
-
A cluster of euhedral feldspar crystals with a schorl crystal
-
First X-ray view of Martian soil—feldspar, pyroxenes, olivine revealed (Curiosity rover at "Rocknest", October 17, 2012)[42]
-
Lunar ferrous anorthosite #60025 (plagioclase feldspar). Collected by Apollo 16 from the Lunar Highlands near Descartes Crater. This sample is currently on display at the National Museum of Natural History in Washington, D.C.
See also
[edit]- List of minerals – List of minerals with Wikipedia articles
- List of countries by feldspar production
- Rainbow lattice sunstone
References
[edit] This article incorporates public domain material from Feldspar and nepheline syenite (PDF). United States Geological Survey.
This article incorporates public domain material from Feldspar and nepheline syenite (PDF). United States Geological Survey.
- ^ Warr, L.N. (2021). "IMA–CNMNC approved mineral symbols". Mineralogical Magazine. 85 (3): 291–320. Bibcode:2021MinM...85..291W. doi:10.1180/mgm.2021.43. S2CID 235729616.
- ^ "Feldspar". Gemology Online. Retrieved 8 November 2012.
- ^ a b Neuendorf, K.K.E.; Mehl, J.P. Jr.; Jackson, J.A., eds. (2005). Glossary of Geology (5th ed.). Alexandria, Virginia: American Geological Institute. p. 232. ISBN 978-0922152896.
- ^ a b Deer, W.A; Howie, R.A.; Zussman, J. (2001). Rock-forming Minerals (2nd edition) Volume 4A. London: Geological Society of London. p. 2. ISBN 1-86239-081-9.
- ^ Anderson, Robert S.; Anderson, Suzanne P. (2010). Geomorphology: The Mechanics and Chemistry of Landscapes. Cambridge University Press. p. 187. ISBN 9781139788700.
- ^ Rudnick, R. L.; Gao, S. (2003). "Composition of the Continental Crust". In Holland, H. D.; Turekian, K. K. (eds.). Treatise on Geochemistry. Vol. 3. New York: Elsevier Science. pp. 1–64. Bibcode:2003TrGeo...3....1R. doi:10.1016/B0-08-043751-6/03016-4. ISBN 978-0-08-043751-4.
- ^ TROLL, V. R. (2002-02-01). "Magma Mixing and Crustal Recycling Recorded in Ternary Feldspar from Compositionally Zoned Peralkaline Ignimbrite A', Gran Canaria, Canary Islands". Journal of Petrology. 43 (2): 243–270. Bibcode:2002JPet...43..243T. doi:10.1093/petrology/43.2.243. ISSN 1460-2415.
- ^ "Metamorphic Rocks." Metamorphic Rocks Information Archived 2007-07-01 at the Wayback Machine. Retrieved on July 18, 2007
- ^ Blatt, Harvey and Tracy, Robert J. (1996) Petrology, Freeman, 2nd ed., pp. 206–210 ISBN 0-7167-2438-3
- ^ "Weathering and Sedimentary Rocks." Geology. Archived 2007-07-03 at the Wayback Machine Retrieved on July 18, 2007.
- ^ Hans Lüschen (1979), Die Namen der Steine. Das Mineralreich im Spiegel der Sprache (2nd ed.), Thun: Ott Verlag, p. 215, ISBN 3-7225-6265-1
- ^ Harper, Douglas. "feldspar". Online Etymology Dictionary. Retrieved 2008-02-08.
- ^ "spar". Oxford English Dictionary. Oxford Dictionaries. Archived from the original on September 26, 2016. Retrieved 13 January 2018.
- ^ a b c d e Feldspar. What is Feldspar? Industrial Minerals Association. Retrieved on July 18, 2007.
- ^ Le Bas, M. J.; Streckeisen, A. L. (1991). "The IUGS systematics of igneous rocks". Journal of the Geological Society. 148 (5): 825–833. Bibcode:1991JGSoc.148..825L. CiteSeerX 10.1.1.692.4446. doi:10.1144/gsjgs.148.5.0825. S2CID 28548230.
- ^ "Rock Classification Scheme - Vol 1 - Igneous" (PDF). British Geological Survey: Rock Classification Scheme. 1: 1–52. 1999. Archived (PDF) from the original on 2022-10-09.
- ^ Philpotts, Anthony R.; Ague, Jay J. (2009). Principles of igneous and metamorphic petrology (2nd ed.). Cambridge, UK: Cambridge University Press. pp. 139–143. ISBN 9780521880060.
- ^ Bowen, N.L. (1956). The Evolution of the Igneous Rocks. Canada: Dover. pp. 60–62.
- ^ Klein, Cornelis; Hurlbut, Cornelius S. Jr. (1993). Manual of mineralogy : (after James D. Dana) (21st ed.). New York: Wiley. p. 559. ISBN 047157452X.
- ^ a b "The Mineral Orthoclase". Feldspar Amethyst Galleries, Inc. Retrieved on February 8, 2008.
- ^ a b "Sanidine Feldspar". Feldspar Amethyst Galleries, Inc. Retrieved on February 8, 2008.
- ^ "Microcline Feldspar". Feldspar Amethyst Galleries, Inc. Retrieved on February 8, 2008.
- ^ Klein & Hurlbut 1993, pp. 532–536.
- ^ Ralph, Jolyon and Chou, Ida. "Perthite". Perthite Profile on mindat.org. Retrieved on February 8, 2008.
- ^ "Buddingtonite".
- ^ "Feldspar Group". mindat.org. Retrieved 4 July 2021.
- ^ Celsian–orthoclase series on Mindat.org.
- ^ Celsian–hyalophane series on Mindat.org.
- ^ Klein & Hurlbut 1993, pp. 533–534.
- ^ a b Blum, Alex E. (1994), Parsons, Ian (ed.), "Feldspars in Weathering", Feldspars and their Reactions, NATO ASI Series, Dordrecht: Springer Netherlands, pp. 595–630, doi:10.1007/978-94-011-1106-5_15, ISBN 978-94-011-1106-5, retrieved 2020-11-18
- ^ Hefferan, Kevin; O'Brien, John (2010). Earth Materials. Wiley-Blackwell. pp. 336–337. ISBN 978-1-4443-3460-9.
- ^ a b c Nelson, Stephen A. (Fall 2008). "Weathering & Clay Minerals". Professor's lecture notes (EENS 211, Mineralogy). Tulane University. Retrieved 2008-11-13.
- ^ Earle, Steven (September 2015). "5.2 Chemical Weathering". Physical Geology. BCcampus.
- ^ a b "Arkose". www.mindat.org. Retrieved 2020-11-18.
- ^ Apodaca, Lori E. Feldspar and nepheline syenite, USGS 2008 Minerals Yearbook
- ^ a b 'Industrial Minerals & Rocks - Commodities, Markets And Uses' J. E. Kogel. Society For Mining, Metallurgy And Exploration, 2006. Pg. 458
- ^ 'Industrial Ceramics' F.Singer, S.S.Singer. Chapman & Hall, 1971
- ^ Neufeld, Rob (4 August 2019). "Visiting Our Past: Feldspar mining and racial tensions". Asheville Citizen-Times. Retrieved 4 August 2019.
- ^ Feldspar And Nepheline Syenite. U.S. Geological Survey, Mineral Commodity Summaries, January 2020
- ^ 'Feldspar Facts' A. Sugden. Ceramic Review, Issue 207, May/June 2004
- ^ Nasa's Curiosity rover finds 'unusual rock'. (12 October 2012) BBC News.
- ^ Brown, Dwayne (October 30, 2012). "NASA Rover's First Soil Studies Help Fingerprint Martian Minerals". NASA. Archived from the original on June 3, 2016. Retrieved October 31, 2012.
Further reading
[edit]- Bonewitz, Ronald Louis (2005). Rock and Gem. New York: DK Publishing. ISBN 978-0-7566-3342-4.
External links
[edit] Media related to Feldspar at Wikimedia Commons
Media related to Feldspar at Wikimedia Commons- Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica. Vol. 10 (11th ed.). Cambridge University Press. p. 245.





![First X-ray view of Martian soil—feldspar, pyroxenes, olivine revealed (Curiosity rover at "Rocknest", October 17, 2012)[42]](/uploads/wikipedia/commons/thumb/1/10/PIA16217-MarsCuriosityRover-1stXRayView-20121017.jpg/126px-PIA16217-MarsCuriosityRover-1stXRayView-20121017.jpg?auto=webp)
