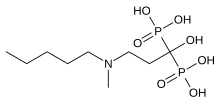
Ibandronic acid
 | |
| Clinical data | |
|---|---|
| Trade names | Boniva, Bonviva, Bondronat, others |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 0.6% |
| Protein binding | 90.9 to 99.5% (concentration-dependent) |
| Metabolism | Nil |
| Elimination half-life | 10 to 60 hours |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.214.537 |
| Chemical and physical data | |
| Formula | C9H23NO7P2 |
| Molar mass | 319.231 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Ibandronic acid is a bisphosphonate medication used in the prevention and treatment of osteoporosis and metastasis-associated skeletal fractures in people with cancer.[4] It may also be used to treat hypercalcemia (elevated blood calcium levels). It is typically formulated as its sodium salt ibandronate sodium.[medical citation needed]
It was patented in 1986 by Boehringer Mannheim and approved for medical use in 1996.[5]
Medical uses
[edit]Ibandronate is indicated for the treatment and prevention of osteoporosis in post-menopausal women.[6] In May 2003, the US Food and Drug Administration (FDA) approved ibandronate as a daily treatment for post-menopausal osteoporosis.[medical citation needed] The basis for this approval was a three-year, randomized, double-blind, placebo-controlled trial women with post-menopausal osteoporosis.[medical citation needed] Each participant also received daily oral doses of calcium and 400IUs [international units] of vitamin D.[medical citation needed] At the study's conclusion, both doses significantly reduced the occurrence risk of new vertebral fractures by 50–52 percent when compared to the effects of the placebo drug.[medical citation needed]
Ibandronate is efficacious for the prevention of metastasis-related bone fractures in multiple myeloma, breast cancer, and certain other cancers.[7]
Adverse effects
[edit]In 2008, the US Food and Drug Administration (FDA) issued a communication warning of the possibility of severe and sometimes incapacitating bone, joint or muscle pain.[8] A study conducted by the American Society of Bone and Mineral Research concluded that long-term use of bisphosphonates, including Boniva, may increase the risk of a rare but serious fracture of the femur.[9] The drug also has been associated with osteonecrosis of the jaw, a relatively rare but serious condition.[10]
Pharmacology
[edit]Mechanism of action
[edit]Nitrogen containing bisphosphonates, which include ibandronate, pamidronate and alendronate exert their effects on osteoclasts mainly by inhibiting the synthesis of isoprenoid lipids such as isopentenyl diphosphate (IPP), farnesyl diphosphate (FPP), and geranylgeranyl diphosphate (GGPP) via the mevalonate pathway. These isoprenoids are used in posttranslational modifcation(prenylation) of small GTPases such as Ras, Rho, and Rac. These prenylated GTPases are necessary for various cellular processes including osteoclast morphology, endosome trafficking, and apoptosis.[11]
| Bisphosphonate | Relative potency |
|---|---|
| Etidronate | 1 |
| Tiludronate | 10 |
| Pamidronate | 100 |
| Alendronate | 100-500 |
| Ibandronate | 500-1000 |
| Risedronate | 1000 |
| Zoledronate | 5000 |
Society and culture
[edit]Brand names
[edit]Ibandronic acid is sold under the brand names Boniva, Bondronat, Bonviva, Bandrone, Ibandrix, Adronil, Bondrova, Bonprove, and Fosfonat.
References
[edit]- ^ "Bondronat EPAR". European Medicines Agency (EMA). 25 June 1996. Retrieved 28 August 2024.
- ^ "Bonviva EPAR". European Medicines Agency (EMA). 23 February 2004. Retrieved 28 August 2024.
- ^ "Iasibon EPAR". European Medicines Agency (EMA). 21 January 2011. Retrieved 30 August 2024.
- ^ Bauss F, Schimmer RC (March 2006). "Ibandronate: the first once-monthly oral bisphosphonate for treatment of postmenopausal osteoporosis". Therapeutics and Clinical Risk Management. 2 (1): 3–18. PMC 1661644. PMID 18360577.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 523. ISBN 9783527607495.
- ^ "Boniva". The American Society of Health-System Pharmacists. Retrieved 3 April 2011.
- ^ Sittig HB (2012). "Pathogenesis and bisphosphonate treatment of skeletal events and bone pain in metastatic cancer: focus on ibandronate". Onkologie. 35 (6): 380–7. doi:10.1159/000338947. PMID 22722461. S2CID 8413102.
- ^ "Information for Healthcare Professionals: Bisphosphonates (marketed as Actonel, Actonel+Ca, Aredia, Boniva, Didronel, Fosamax, Fosamax+D, Reclast, Skelid, and Zometa)". U.S. Food and Drug Administration. Retrieved 27 October 2010.
- ^ Shane E, Burr D, Ebeling PR, Abrahamsen B, Adler RA, Brown TD, et al. (American Society for Bone and Mineral Research) (November 2010). "Atypical subtrochanteric and diaphyseal femoral fractures: report of a task force of the American Society for Bone and Mineral Research". Journal of Bone and Mineral Research. 25 (11): 2267–2294. doi:10.1002/jbmr.253. PMID 20842676. S2CID 15194275.
{{cite journal}}: CS1 maint: overridden setting (link); Lay summary in: "JBMR Publishes ASBMR Task Force Report on Atypical Femoral Fractures". Journal of Bone and Mineral Research. 14 September 2010. Archived from the original on 9 April 2016. Retrieved 27 October 2010. - ^ "Osteonecrosis of the jaw (ONJ) and drug treatments for osteoporosis" (PDF). United Kingdom: The National Osteoporosis Society. Archived from the original (PDF) on 17 June 2017. Retrieved 14 August 2018.
- ^ Rogers MJ, Gordon S, Benford HL, Coxon FP, Luckman SP, Monkkonen J, et al. (June 2000). "Cellular and molecular mechanisms of action of bisphosphonates". Cancer. 88 (12 Suppl): 2961–2978. doi:10.1002/1097-0142(20000615)88:12+<2961::AID-CNCR12>3.0.CO;2-L. PMID 10898340.
- ^ Tripathi KD (30 September 2013). Essentials of Medical Pharmacology (Seventh ed.). New Delhi: Jaypee Brothers Medical Publisher. ISBN 9789350259375. OCLC 868299888.
