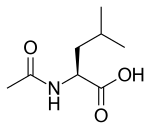
Levacetylleucine
 | |
| Clinical data | |
|---|---|
| Trade names | Aqneursa |
| Other names | IB1001 |
| AHFS/Drugs.com | Aqneursa |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.013.370 |
| Chemical and physical data | |
| Formula | C8H15NO3 |
| Molar mass | 173.212 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Levacetylleucine, sold under the brand name Aqneursa, is a medication used for the treatment of neurological manifestations of Niemann-Pick disease type C.[1][2] Levacetylleucine is a modified version of the amino acid leucine (N-Acetyl-L-Leucine).[1] It is taken by mouth.[1]
The most common side effects include abdominal pain, difficulty swallowing, upper respiratory tract infections, and vomiting.[1][2]
Levacetylleucine was approved for medical use in the United States in September 2024.[1][2][3] Levacetylleucine is the second medication approved by the US Food and Drug Administration (FDA) for the treatment of Niemann-Pick disease type C.[2]
Medical uses
[edit]Levacetylleucine is indicated for the treatment of neurological manifestations of Niemann-Pick disease type C in people weighing at least 15 kilograms (33 lb).[1][2]
Adverse effects
[edit]The most common side effects include abdominal pain, difficulty swallowing, upper respiratory tract infections, and vomiting.[2]
Levacetylleucine may cause embryo-fetal harm if used during pregnancy.[1][2]
History
[edit]The safety and efficacy of levacetylleucine for the treatment of Niemann-Pick disease type C were evaluated in a randomized, double-blind, placebo-controlled, two-period, 24-week crossover study.[2] The duration was twelve weeks for each treatment period.[2] The study enrolled 60 participants.[2] To be eligible for the study participants had to be four years of age or older with a confirmed diagnosis of Niemann-Pick disease type C and at least mild disease-related neurological symptoms.[2] Participants could receive miglustat, an enzyme inhibitor, as background treatment in the study.[2]
The US Food and Drug Administration (FDA) granted the application for levacetylleucine priority review, fast track, orphan drug, and rare pediatric disease designations.[2] The FDA granted approval of Aqneursa to IntraBio Inc.[2]
Society and culture
[edit]Legal status
[edit]Levacetylleucine was approved for medical use in the United States in September 2024.[1][2][4]
Names
[edit]Levacetylleucine is the international nonproprietary name.[5]
Research
[edit]N-Acetyl-L-Leucine is being studied for the treatment of GM2 gangliosidoses (Tay-Sachs and Sandhoff diseases)[6] and ataxia-telangiectasia.[7]
References
[edit]- ^ a b c d e f g h i "Aqneursa- levacetylleucine granule, for suspension". DailyMed. 24 September 2024. Retrieved 5 October 2024.
- ^ a b c d e f g h i j k l m n o "FDA Approves New Drug to Treat Niemann-Pick Disease, Type C". U.S. Food and Drug Administration (Press release). 24 September 2024. Retrieved 25 September 2024.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "IntraBio Announces U.S. FDA Approval of Aqneursa for the Treatment of Niemann-Pick Disease Type C". IntraBio (Press release). 25 September 2024. Retrieved 26 September 2024.
- ^ "Novel Drug Approvals for 2024". U.S. Food and Drug Administration (FDA). 1 October 2024. Retrieved 29 November 2024.
- ^ World Health Organization (2024). "International nonproprietary names for pharmaceutical substances (INN): proposed INN: list 131". WHO Drug Information. 38 (2). hdl:10665/378367. ISBN 9789240098558.
- ^ Martakis K, Claassen J, Gascon-Bayari J, Goldschagg N, Hahn A, Hassan A, et al. (March 2023). "Efficacy and Safety of N-Acetyl-l-Leucine in Children and Adults With GM2 Gangliosidoses". Neurology. 100 (10): e1072 – e1083. doi:10.1212/WNL.0000000000201660. PMC 9990862. PMID 36456200.
- ^ Fields T, Patterson M, Bremova-Ertl T, Belcher G, Billington I, Churchill GC, et al. (January 2021). "A master protocol to investigate a novel therapy acetyl-L-leucine for three ultra-rare neurodegenerative diseases: Niemann-Pick type C, the GM2 gangliosidoses, and ataxia telangiectasia". Trials. 22 (1): 84. doi:10.1186/s13063-020-05009-3. PMC 7821839. PMID 33482890.
Further reading
[edit]- Churchill GC, Strupp M, Factor C, Bremova-Ertl T, Factor M, Patterson MC, et al. (August 2021). "Acetylation turns leucine into a drug by membrane transporter switching". Scientific Reports. 11 (1): 15812. Bibcode:2021NatSR..1115812C. doi:10.1038/s41598-021-95255-5. PMC 8338929. PMID 34349180.
- Bremova-Ertl T, Ramaswami U, Brands M, Foltan T, Gautschi M, Gissen P, et al. (February 2024). "Trial of N-Acetyl-l-Leucine in Niemann-Pick Disease Type C". The New England Journal of Medicine. 390 (5): 421–431. doi:10.1056/NEJMoa2310151. PMID 38294974.
- Tifft CJ (February 2024). "N-Acetyl-l-Leucine and Neurodegenerative Disease". The New England Journal of Medicine. 390 (5): 467–470. doi:10.1056/NEJMe2313791. PMID 38294981.
External links
[edit]- Clinical trial number NCT05163288 for "A Pivotal Study of N-Acetyl-L-Leucine on Niemann-Pick Disease Type C" at ClinicalTrials.gov
