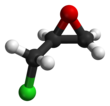
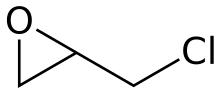
Epichlorohydrin
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-(Chloromethyl)oxirane | |||
| Other names
(Chloromethyl)oxirane
Epichlorohydrin 1-Chloro-2,3-epoxypropane γ-Chloropropylene oxide Glycidyl chloride ECH | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 79785 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.003.128 | ||
| EC Number |
| ||
| 164180 | |||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 2023 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H5ClO | |||
| Molar mass | 92.52 g/mol | ||
| Appearance | colorless liquid | ||
| Odor | garlic or chloroform-like | ||
| Density | 1.1812 g/cm3 | ||
| Melting point | −25.6 °C (−14.1 °F; 247.6 K) | ||
| Boiling point | 117.9 °C (244.2 °F; 391.0 K) | ||
| 7% (20°C)[2] | |||
| Vapor pressure | 13 mmHg (20°C)[2] | ||
| Hazards | |||
| GHS labelling: | |||
    
| |||
| Danger | |||
| H226, H301, H311, H314, H317, H331, H350 | |||
| P201, P202, P210, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P272, P280, P281, P301+P310, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P310, P311, P312, P321, P322, P330, P333+P313, P361, P363, P370+P378, P403+P233, P403+P235, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 32 °C (90 °F; 305 K) | ||
| Explosive limits | 3.8–21%[2] | ||
| Lethal dose or concentration (LD, LC): | |||
LC50 (median concentration)
|
3617 ppm (rat, 1 hr) 2165 ppm (rat, 1 hr) 250 ppm (rat, 8 hr) 244 ppm (rat, 8 hr) 360 ppm (rat, 6 hr)[3] | ||
LCLo (lowest published)
|
250 ppm (rat, 4 hr)[3] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 5 ppm (19 mg/m3) [skin][2] | ||
REL (Recommended)
|
Carcinogen[2] | ||
IDLH (Immediate danger)
|
Ca [75 ppm][2] | ||
| Safety data sheet (SDS) | External MSDS | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Epichlorohydrin (abbreviated ECH) is an organochlorine compound and an epoxide. Despite its name, it is not a halohydrin. It is a colorless liquid with a pungent, garlic-like odor, moderately soluble in water, but miscible with most polar organic solvents.[4] It is a chiral molecule generally existing as a racemic mixture of right-handed and left-handed enantiomers. Epichlorohydrin is a highly reactive electrophilic compound and is used in the production of glycerol, plastics, epoxy glues and resins, epoxy diluents and elastomers.
Production
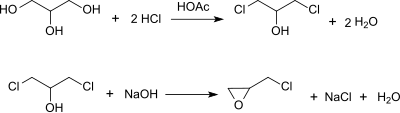
[edit]Epichlorohydrin is traditionally manufactured from allyl chloride in two steps, beginning with the addition of hypochlorous acid, which affords a mixture of two isomeric alcohols:[5][6]
In the second step, this mixture is treated with base to give the epoxide:
In this way, more than 800,000 tons (1997) of epichlorohydrin are produced annually.[7]
Glycerol routes
[edit]Epichlorohydrin was first described in 1848 by Marcellin Berthelot. The compound was isolated during studies on reactions between glycerol and gaseous hydrogen chloride.[8]
Reminiscent of Berthelot's experiment, glycerol-to-epichlorohydrin (GTE) plants have been commercialized. This technology capitalizes on the availability of cheap glycerol from biofuels processing.[9] In the process developed by Dow Chemical, glycerol undergoes two substitution reactions when treated with hydrogen chloride in the presence of a carboxylic acid catalyst. This is the same intermediate formed in the allyl chloride/hypochlorous acid process, and is likewise then treated with base to form epichlorohydrin.[10]
Other routes
[edit]Routes that involve fewer chlorinated intermediates have continued to attract interest. One such process entails epoxidation of allyl chloride.[11]
Applications
[edit]Glycerol and epoxy resins synthesis
[edit]Epichlorohydrin is mainly converted to bisphenol A diglycidyl ether, a building block in the manufacture of epoxy resins.[12] It is also a precursor to monomers for other resins and polymers. Another usage is the conversion to synthetic glycerol. However, the rapid increase in biodiesel production, where glycerol is a waste product, has led to a glut of glycerol on the market, rendering this process uneconomical. Synthetic glycerol is now used only in sensitive pharmaceutical, and biotech applications where quality standards are very high.[13]
Minor and niche applications
[edit]Epichlorohydrin is a versatile precursor in the synthesis of many organic compounds. For example, it is converted to glycidyl nitrate, an energetic binder used in explosive and propellant compositions.[14] The epichlorohydrin is reacted with an alkali nitrate, such as sodium nitrate, producing glycidyl nitrate and alkali chloride. It is used as a solvent for cellulose, resins, and paints, and it has found use as an insect fumigant.[15]
Polymers made from epichlorohydrin, e.g., polyamide-epichlorohydrin resins, are used in paper reinforcement and in the food industry to manufacture tea bags, coffee filters, and sausage/salami casings as well as with water purification.[16]
An important biochemical application of epichlorohydrin is its use as crosslinking agent for the production of Sephadex size-exclusion chromatographic resins from dextrans.[17]
Safety
[edit]Epichlorohydrin is classified by several international health research agencies and groups as a probable or likely carcinogen in humans.[18][19][20] Prolonged oral consumption of high levels of epichlorohydrin could result in stomach problems and an increased risk of cancer.[21] Occupational exposure to epichlorohydrin via inhalation could result in lung irritation and an increased risk of lung cancer.[22]
References
[edit]- ^ Merck Index, 12th Edition, 3648.
- ^ a b c d e f NIOSH Pocket Guide to Chemical Hazards. "#0254". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "Epichlorohydrin". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ "EPA consumer factsheet". Epa.gov. Retrieved 2011-12-02.
- ^ Braun, G. (1936). "Epichlorohydrin and Epybromohydrin". Organic Syntheses. 16: 30. doi:10.15227/orgsyn.016.0030.
- ^ Guenter Sienel; Robert Rieth; Kenneth T. Rowbottom. "Epoxides". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a09_531. ISBN 978-3527306732.
- ^ Ludger Krähling; Jürgen Krey; Gerald Jakobson; Johann Grolig; Leopold Miksche. "Allyl Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a01_425. ISBN 978-3527306732.
- ^ Berthelot, Marcellin (1854). "Sur les combinaisons de la glycérine avec les acides et sur la synthèse des principes immédiats des graisses animaux". Ann. Chim. Phys. Série 3. 41: 216–319. Archived from the original on 2015-04-02. Retrieved 2015-03-02.
- ^ Doris de Guzman (2011-01-20). "Growing glycerine-to-ECH plants". ICIS Green Chemicals. Archived from the original on 2012-04-19. Retrieved 2012-03-05.
- ^ Bell, Bruce M.; Briggs, John R.; Campbell, Robert M.; Chambers, Susanne M.; Gaarenstroom, Phil D.; Hippler, Jeffrey G.; Hook, Bruce D.; Kearns, Kenneth; et al. (2008). "Glycerin as a Renewable Feedstock for Epichlorohydrin Production. The GTE Process" (PDF). CLEAN - Soil, Air, Water. 36 (8): 657. Bibcode:2008CSAW...36..657B. doi:10.1002/clen.200800067. Archived from the original (full text reprint) on 2012-07-18. Retrieved 2012-03-05.
- ^ Jun Li; Gongda Zhao; Shuang Gao; Ying Lv; Jian Li; Zuwei Xi (2006). "Epoxidation of Allyl Chloride to Epichlorohydrin by a Reversible Supported Catalyst with H2O2 under Solvent-Free Conditions". Org. Process Res. Dev. 10 (5): 876–880. doi:10.1021/op060108k.
- ^ Pham, Ha Q.; Marks, Maurice J. (2012). "Epoxy Resins". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a09_547.pub2. ISBN 978-3-527-30673-2.
- ^ Taylor, Phil (16 October 2008). "Synthetic glycerine is back (but never really went away)!". In-Pharma Technologist. Retrieved 29 November 2018.[permanent dead link]
- ^ Gould, R.F. Advanced Propellant Chemistry, ACS Chemistry Series 54, 1966
- ^ "Suburban Water Testing Labs:Epichlorohydrin Fact Sheet". H2otest.com. Archived from the original on 2012-04-05. Retrieved 2011-12-02.
- ^ "Government of Canada Chemical Substances: Oxirane,(chloromethyl)-(Epichlorohydrin) CAS Registry Number 106-89-8". 13 February 2008. Retrieved 2013-05-07.
- ^ "Size Exclusion Chromatography | Cytiva (cytivalifesciences.com)". Size Exclusion Chromatography | Cytiva (cytivalifesciences.com). cytivalifesciences.com. Retrieved 2024-07-01.
- ^ "EPA Integrated Risk Information System: Epichlorohydrin (CASRN 106-89-8)". Retrieved 2013-05-07.
- ^ "Government of Canada: Screening Assessment for Epichlorohydrin". 27 January 2010. Retrieved 2013-05-07.
- ^ "NIOSH Pocket Guide to Chemical Hazards - Epichlorohydrin". Retrieved 2013-09-20.
- ^ "Basic Information about Epichlorohydrin in Drinking Water". Retrieved 2013-05-07.
- ^ "Government of Canada: Screening Assessment for Epichlorohydrin". 27 January 2010. Retrieved 2013-05-07.